UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE 14A
Proxy Statement Pursuant to Section 14(a) of the
Securities Exchange Act of 1934
Filed by the Registrant ☒ | | | Filed by a Party other than the Registrant ☐ |
Check the appropriate box:
| | Preliminary Proxy Statement | ||
☐ | | Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2)) | |
| | Definitive Proxy Statement | ||
☐ | | Definitive Additional Materials | |
☐ | | Soliciting Material Pursuant to §240.14a-12 |
Elanco Animal Health Incorporated
(Name of registrant as specified in its charter)
(Name of person(s) filing proxy statement, if other than the registrant)
Payment of Filing Fee (Check all boxes that apply): | ||||||
☒ | | No fee required. | ||||
☐ | | Fee paid previously with preliminary materials. | ||||
☐ | | Fee computed on table in exhibit required by Item 25(b) per Exchange Act Rules 14a6(i)(1) and 0-11. | ||||

Letter from our Board of
Directors
Dear fellow Elanco shareholders,Shareholders,
Since Elanco’s launch as an independent global animal health leader.
Shareholder-Aligned Governance Enhancements in revenue,Response to Feedback
The Board is committed to listening to and responding to shareholder feedback. In 2022, the company eliminated the supermajority vote requirements to amend the Company’s Articles of Incorporation and Bylaws. As the company continues to progress, in this proxy statement, the Board is seeking shareholder approval to enact further governance enhancements:
| ● Commence a process to declassify the Board of Directors; |
| ● Adopt a majority vote standard for uncontested elections of directors; |
| ● Allow shareholders to amend the Company’s Bylaws; and |
| ● Allow shareholders under certain circumstances to call special meetings of shareholders. |
The Board values our shareholders’ opinions and appreciates their feedback which has informed our actions. In the past year, members of the Board engaged with an Adjusted EBITDA of slightly above $1.0 billion for the full year. While we experienced topline pressure, our team’s focus on operational discipline contributed to 80 basis points of Adjusted EBITDA margin expansion for the year. Our transition to a metric similar to economic valued added, Elanco Cash Earnings, has furthered incentivized our productivity efforts and reinforced a culture of ownership across our nearly 10,000 Elanco employees.
Board Leadership Changes
As announced in January, the most significant launch window in our history, with a pathBoard has selected Lawrence E. Kurzius to six potential blockbuster approvals – these are products with anticipated peak annual sales of at least $100 million – by the first half of 2024, furthering our confidence in our ability to add $600-$700 million in revenue from our innovation pipeline by 2025.
The Board of ESG issues is ingrained in all aspects of our business and driven from within our organization: demonstrated by the commitment of our people and the power of our Elanco Healthy Purpose™ platform. Our global reach and strong business position helped us progress toward our 2030 Healthy Purpose commitments. We continue to promote the health and well-being of animals, people, the planet, and our enterprise.

Sincerely,
Elanco Board of Directors I

Letter from our CEO
Dear Shareholders,
In 2023, Elanco demonstrated progress and momentum on our Innovation, Portfolio and Productivity (IPP) strategy to deliver long-term shareholder value. We returned to revenue growth and made meaningful progress on our pipeline of potential blockbuster products, while completing our ERP system integration, a significant milestone that will significantly improve free cash flow for debt pay down. Since our IPO in 2018, we have expanded our portfolio and scale through strategic acquisitions, positioned Elanco as the omnichannel Pet Health leader around the globe and are now well-positioned to deliver on our next era of innovation and growth.
Building Portfolio Momentum
In the second half of 2023, Elanco returned to growth, increasing revenues by 5% at constant currency for two consecutive quarters, ending the full year at 1% constant currency growth. As we stabilize our base portfolio, both pet health and farm animal performance improved in 2023 with price contributing across the business. As the pet health omnichannel leader, Elanco is well positioned to maximize our offering, complementing our long-standing veterinary relationships with expanding over the counter offerings in pet retail and e-commerce. We are able to meet the pet owner wherever they want to thankshop for pet care—from the veterinarian to the neighborhood store to the convenience of their couch – at a wide variety of price points.
Investing in High Impact Innovation
We made meaningful progress advancing our innovative, late-stage product pipeline. We have six potential blockbuster products, each with anticipated annual revenue of more than $100 million over time, that are expected to enter the U.S. market by 2025. Two of these products are already in the market and contributed to more than doubling our growth from new products in 2023. We look forward to bringing the additional products to market in dermatology and parasiticides for dogs and methane reduction for cattle. We are also advancing a portfolio of development projects with expected differentiated profiles and blockbuster potential that we believe have the opportunity to contribute meaningful growth and earnings in the second half of the decade. Additionally, we invested in important commercial capabilities and expanded share of voice that we believe will maximize the value of our current portfolio and expected launches in 2024 and beyond.
Finally, we are pleased with efforts to pioneer the livestock sustainability market, supporting the development of the first livestock carbon inset marketplace and initial sale of carbon credits in the U.S. We see long-term potential to support our customers and help the industry reduce its climate impact while generating a meaningful new revenue stream for producers.
Improving Productivity and Cash Flow
We continue to focus the global organization on prioritizing cash generation aligned with our Elanco Cash Earnings, EVA-like corporate bonus program. In 2023, we paid down $76 million in gross debt and finished the year with a net leverage ratio of 5.6x. We also achieved the significant milestone of completing our ERP system integration, which will free up more than $100 million of free cash flow for debt paydown in 2024. We are taking deliberate steps to concentrate our focus on areas of higher earnings potential in pet health and livestock sustainability and accelerate deleveraging. In February 2024, we announced an agreement to divest our aquaculture business for $1.3 billion, approximately 7.4x the estimated 2023 revenue. We expect the proceeds of this transaction, plus our anticipated improving free cash flow, to be used for debt paydown and improve our leverage profile, moving toward a high 3x to low 4x net debt to adjusted EBITDA ratio by the end of 2025.
Fostering an Engaged, Ownership-Minded Workforce to Drive Value
With our transition to Elanco Cash Earnings, our teams around the world are focused on shifting resources to opportunities of greatest growth and earnings potential. We are focused on advancing our IPP strategy to deliver on three priorities: sustained revenue growth, delivering a robust innovation pipeline and improved cash conversion. Progress and momentum against these priorities are expected to fuel long-term sustainable growth, offering a compelling value proposition.
Additionally, we prioritize attracting, retaining, and developing a workforce with the skills and diversity of experience to innovate and bring new products to market. Ultimately, our team is grounded in the passionate pursuit of our purpose. For the past seven decades the Elanco team has been guided by a deep collective belief – going beyond for our customers makes life better for animals, making life better.
Thank you for your investment in Elanco.
Jeff Simmons
President and incorporating your feedback into our discussions as we enter the most significant launch window in our history.Chief Executive Officer
Preliminary Copy – Subject to Completion

R. David Hoover,



of Shareholders of ELANCO ANIMAL HEALTH INCORPORATED, an Indiana corporation, willto be a virtual meeting of shareholders, conducted via live audio webcast at www.virtualshareholdermeeting.com/ELAN2023 Held
on Thursday, May 18, 2023, at 8:00 a.m., Eastern Time, to consider and act upon the following matters:30, 2024
ELANCO ANIMAL HEALTH INCORPORATED 2500 Innovation Way Greenfield, Indiana 46140 | The 2024 Annual Meeting of Voting Matters: | ||
Date & Time 12:00 p.m., Eastern Time, Thursday, May 30, 2024
Location Audio webcast at: virtualshareholdermeeting.
Record Date Close of business on April 11, 2024 | 1 | Election of | |
Ratification of the appointment of Ernst & Young LLP as Elanco’s independent registered public accounting firm for 2024. | |||
Advisory vote on the compensation of Elanco’s named executive officers. | |||
| Management proposal to amend Elanco’s Articles of Incorporation to provide shareholders with the right to request special meetings of shareholders. | |||
| In addition, we will transact such other business as may properly come before the meeting. | |||
This Notice and the accompanying Proxy Statement, or a Notice of Internet Availability of Proxy Materials, are expected to be mailed to shareholders commencing April [•], 2024. By Order of the Board of Directors, Shiv O'Neill Executive Vice President, General Counsel and Corporate Secretary April [•], 2024 | |||

Executive Vice President, General Counsel and Corporate SecretaryVoting
Even though you may plan to participate in the meeting online, please vote by telephone or the Internet, or execute the proxy card and mail it promptly. Telephone and Internet voting information is provided on the notice mailed to you or in this Proxy Statement. If you participate in the virtual meeting, you may revoke your proxy and vote your shares electronically during the meeting.
The Notice of 20232024 Annual Shareholders Meeting, Proxy Statement and Annual Report on Form 10-K are available at www.proxyvote.com.www.proxyvote.com.
Table of
Contents
WAYS TO VOTE
Online Prior to the Annual Meeting You may vote by proxy by visiting www.proxyvote.com and entering the control number found on your Notice of Internet Availability.
Online During the Annual Meeting You may vote online during the Annual Meeting by visiting www.virtualshareholder meeting.com/ELAN2024
Telephone If you request printed copies of the proxy materials by mail, you will receive a proxy card or voting instruction form and you may vote by proxy by calling the toll-free number found on the card or form.
If you request printed copies of the proxy materials by mail, you will receive a proxy card or voting instruction form. | This summary highlights information contained in this Proxy Statement. This summary does not contain all of the information that you should consider and you should carefully read the entire Proxy Statement before voting.
Voting Matters and Recommendations
| ||||
| Item of Business | Board Recommendation | Page | |||
| 1 | Election of four director nominees to serve three-year terms. | “FOR” Each Nominee | 9 | ||
| 2 | Ratification of the appointment of Ernst & Young LLP as Elanco’s independent registered public accounting firm for 2024. | “FOR” | 47 | ||
| 3 | Advisory vote on the compensation of Elanco’s named executive officers. | “FOR” | 50 | ||
| 4 | Management proposal to amend Elanco’s Articles of Incorporation to declassify the Board of Directors. | “FOR” | 87 | ||
| 5 | Management proposal to amend Elanco’s Articles of Incorporation to adopt a majority vote standard for uncontested elections of directors. | “FOR” | 89 | ||
| 6 | Management proposal to amend Elanco’s Articles of Incorporation to provide shareholders with the right to amend our Bylaws. | “FOR” | 91 | ||
| 7 | Management proposal to amend Elanco’s Articles of Incorporation to provide shareholders with the right to request special meetings of shareholders. | “FOR” | 93 | ||
Items of Business | | | Board Recommendation | | | Page | |||
1 | | | Election of the four director nominees to serve three-year terms. | | | “FOR” Each Nominee | | | |
2 | | | Ratification of the appointment of Ernst & Young LLP as Elanco’s independent registered public accounting firm for 2023. | | | “FOR” | | | |
3 | | | Advisory vote on the compensation of Elanco’s named executive officers. | | | “FOR” | | | |
4 | | | Approval of the Amended and Restated Elanco Animal Health Incorporated Employee Stock Purchase Plan. | | | “FOR” | | | |
5 | | | Approval of the Amended and Restated 2018 Elanco Animal Health Incorporated Stock Plan. | | | “FOR” | | | |
 |
| | | | Committees of the Board | | |||||||||||||||||||||||||
| | Name | | | Primary Occupation | | | Age | | | Director Since | | | Independent | | | AC | | | CHCC | | | FOC | | | ISTC | | | CGC | |
| | Class II Director Nominees – Nominees for Election at the 2023 Annual Meeting for Terms Expiring in 2026 | | | | | | | | | | | | | | | | |||||||||||||
| | Michael J. Harrington | | | Former General Counsel, Lilly | | | 60 | | | 2018 | | |  | | | | | | | | |  | | | | ||||
| | R. David Hoover (Chairman) | | | Former Chairman and CEO, Ball Corporation | | | 77 | | | 2018 | | |  | | | | |  | | | | | | |  | | |||
| | Deborah T. Kochevar | | | Senior Fellow, Fletcher School of Law and Diplomacy, Tufts University | | | 66 | | | 2019 | | |  | | | | | | | | |  | | |  | | |||
| | Kirk P. McDonald | | | CEO, Group M North America | | | 56 | | | 2019 | | |  | | | | |  | | | | |  | | | | |||
| | Class III Directors — Terms Expiring in 2024 | | | | | | | | | | | | | | | | |||||||||||||
| | William F. Doyle | | | Executive Chairman, Novocure Ltd. | | | 60 | | | 2020 | | |  | | | | | | |  | | |  | | | | |||
| | Art A. Garcia | | | Former CFO, Ryder System, Inc. | | | 62 | | | 2019 | | |  | | |  | | | | |  | | | | | | |||
| | Denise Scots-Knight | | | Co-Founder and CEO, Mereo BioPharma Group plc | | | 63 | | | 2019 | | |  | | | | |  | | | | |  | | | | |||
| | Jeffrey N. Simmons | | | President and CEO, Elanco Animal Health Incorporated | | | 55 | | | 2018 | | | | | | | | |  | | | | | | |||||
| | Class I Directors — Terms Expiring in 2025 | | | | | | | | | | | | | | | | |||||||||||||
| | Kapila K. Anand | | | Retired Partner, KPMG LLP | | | 69 | | | 2018 | | |  | | |  | | | | | | | | |  | | |||
| | John P. Bilbrey | | | Former Chairman and CEO, The Hershey Company | | | 66 | | | 2019 | | |  | | |  | | | | |  | | | | | | |||
| | Paul Herendeen | | | Former Chief Financial Officer, Bausch Health | | | 67 | | | 2020 | | |  | | |  | | | | |  | | | | | | |||
| | Lawrence E. Kurzius | | | Chairman & CEO McCormick & Company | | | 65 | | | 2018 | | |  | | | | |  | | | | | | |  | | |||
| | | | | | | | | | | | | | | | | | | | | ||||||||||
| Name | Primary Occupation | Age | Director Since | Independent |
| CLASS I DIRECTORS — Terms Expiring in 2025 | ||||
| Kapila Anand | Retired Partner, KPMG LLP | 70 | 2018 |  |
| John Bilbrey | Former Chairman, CEO and President, The Hershey Company | 67 | 2019 |  |
| Paul Herendeen | Former CFO, Bausch Health Companies, Inc. | 68 | 2020 |  |
| Lawrence Kurzius | Executive Chairman, McCormick & Company, Inc. | 66 | 2018 |  |
| Craig Wallace | President, C.S. Wallace Investments + Strategy | 60 | 2024 |  |
| CLASS II DIRECTORS— Terms Expiring in 2026 | ||||
| Michael Harrington | Former General Counsel, Eli Lilly and Company | 61 | 2018 |  |
| R. David Hoover | Former Chairman and CEO, Ball Corporation | 78 | 2018 |  |
| Deborah Kochevar | Senior Fellow, The Fletcher School of Law and Diplomacy and Dean Emerita, Cummings School of Veterinary Medicine, Tufts University | 67 | 2019 |  |
| Kirk McDonald | Former CEO, Group M North America | 57 | 2019 |  |
| Kathy Turner | Former Senior Vice President and Chief Marketing Officer, IDEXX | 60 | 2024 |  |
| CLASS III DIRECTORS— Director Nominees for Terms Expiring in 2027 | ||||
| William Doyle | Executive Chairman, Novocure Ltd. | 61 | 2020 |  |
| Art Garcia | Former CFO, Ryder System, Inc. | 63 | 2019 |  |
| Denise Scots-Knight | Co-Founder and CEO, Mereo BioPharma Group plc | 64 | 2019 |  |
| Jeffrey Simmons | President and CEO, Elanco Animal Health Incorporated | 56 | 2018 | |
Our Board Highlights
Elanco Animal Health Incorporated | 2 |  |
 |
We are committed to the values of effective corporate governance and high ethical standards. We continue to evolve our Board and our corporate governance practices. Many of our changes have been influenced by the valuable feedback we have received from our shareholders and other stakeholders who provide important external viewpoints that help inform our decisions. For more information about our corporate governance practices, including several enhancements we made in 2022, see “Corporate Governance” beginning on page 2427below.
Independent Oversight | ||||||||||||||
| | • | All directors, including our Board Chairman, are independent, except for our CEO | ||||||||||||
• | Four Board | |||||||||||||
• | Regular executive sessions of independent directors at Board meetings (chaired by the independent Board Chairman) and Committee meetings (chaired by the independent | |||||||||||||
• | Active Board and committee oversight of our strategy and risk management, including ESG-related matters | | ||||||||||||
Board Refreshment and Practices | ||||||||||||||
| | ||||||||||||||
• | Seven new independent directors since 2019, | |||||||||||||
including two in 2024 • | Comprehensive, ongoing Board succession planning process | |||||||||||||
• | Annual Board and committee self-assessments led by the independent Corporate Governance Committee | |||||||||||||
• | Board policy limits director membership on other public company boards | |||||||||||||
• | Continuing director education on key topics and issues • Changes proposed for shareholder approval at the Annual Meeting: - Commence a process to declassify the Board beginning at the 2025 annual meeting of shareholders - Adopt a majority vote standard for uncontested elections of directors - Right of shareholders owning a majority of votes entitled to be cast to amend the Bylaws - Right of shareholders owning at least 25% of common stock to request special meeting of shareholders | | ||||||||||||
Shareholder Rights | ||||||||||||||
| | • | 3%/3 years proxy access right for shareholders, adopted in 2022 | ||||||||||||
• | Shareholders can approve amendments to our Articles of Incorporation and Bylaws with a simple majority vote • One class of outstanding shares with each share entitled to one vote | | ||||||||||||
Governance Practices | ||||||||||||||
| | • | |||||||||||||
Code of Conduct applicable to all employees and directors | ||||||||||||||
• | Corporate Governance Guidelines and Financial Code of Ethics • Clawback policy applicable to executives • Rigorous executive stock ownership requirements • Prohibition on hedging or pledging Elanco stock • Regular review of succession planning for CEO and other key executives | |||||||||||||
• | ||||||||||||||
• | Comprehensive shareholder engagement program with independent director participation | | ||||||||||||
| 3 | 2024 Proxy Statement |  |
Our executive compensation program is designed to help achieve the goals of attracting, engaging and retaining highly talented individuals who are committed to our core values of integrity, excellence and respect for people, while balancing the long-term interests of shareholders and customers. We accomplish this, in part, by delivering senior executive pay with a greater emphasis on equity and lower weighting on cash to promote an ownership mentality and help ensure shareholder alignment.
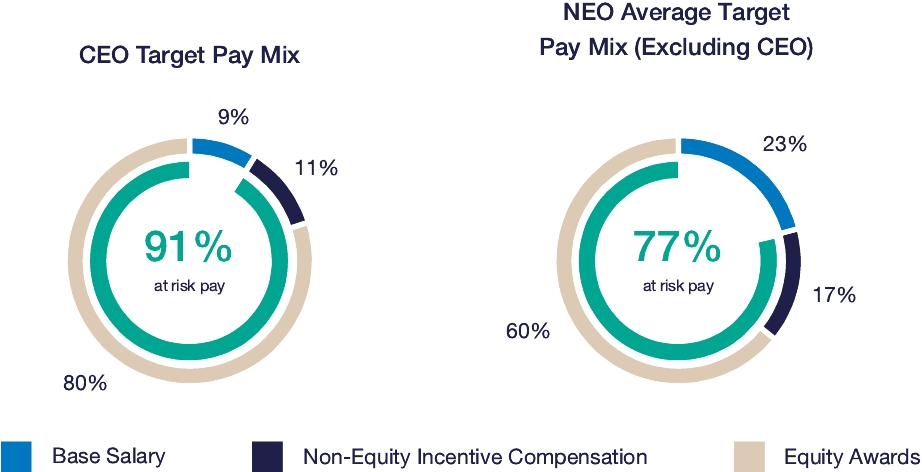
Highlights for 2023 include:
| • | Continued use of Elanco Cash Earnings (ECE) and Adjusted EBITDAR as incentive metrics to focus on productivity efforts, reinforce strong balance sheet management, and align with shareholder interests |
| • | Payouts of both annual cash incentives and performance share awards for performance periods ended in 2023 were both below 80% of target, reflecting a rigorous pay-for-performance discipline |
| • | Compensation program reflected continued mix of compensation elements, with substantial portion of compensation tied to our operating and financial performance and the performance of our stock price, as set forth below: |
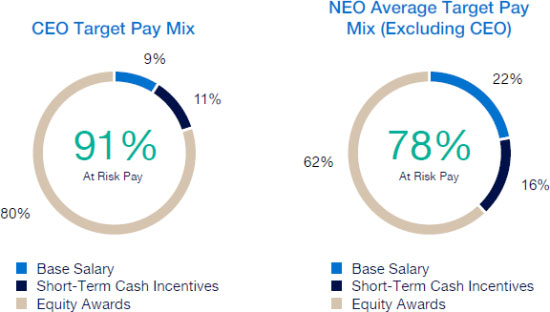
| • | 2023 marked culmination of the “glidepath” trajectory, with all named executive officers now receiving target compensation opportunities at or near peer median levels |
| Elanco Animal Health Incorporated | 4 | 2024 Proxy Statement |  |


| Elanco Animal Health Incorporated | 2024 Proxy Statement |

| Elanco Animal Health Incorporated | 7 | 2024 Proxy Statement |

| Elanco Animal Health Incorporated | 8 | 2024 Proxy Statement |
Proposal No. Election of Directors | ||
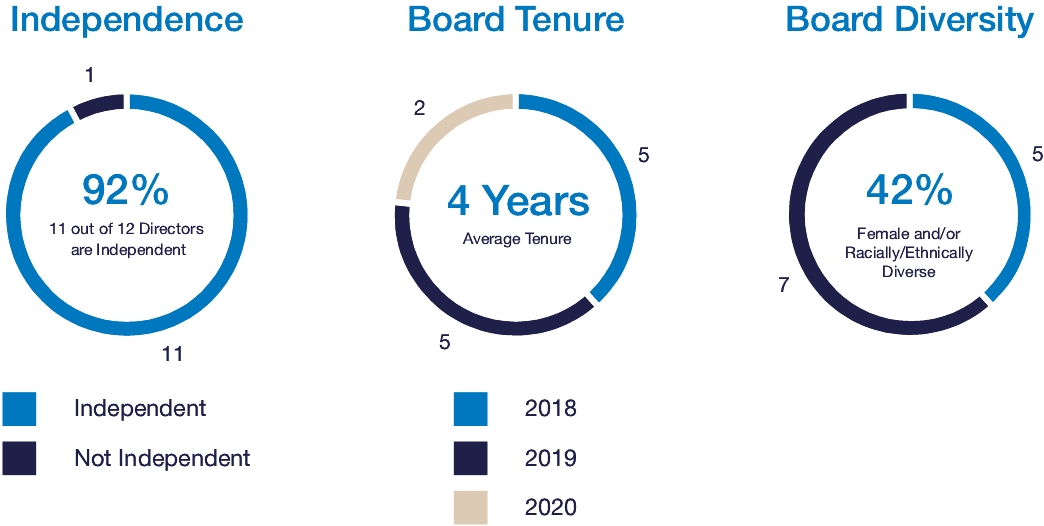
Under our Amended and Restated Articles of Incorporation (our “Articles of Incorporation”), our Board is divided into three classes with approximately one-third of the directors standing for election each year. Our Board currently consists of twelve14 directors. The directors hold office for staggered terms of three years (and until their successors are elected and qualified, or until their earlier death, resignation, or removal). One of the three classes is elected each year to succeed the directors whose terms are expiring.
The “Class II”Class III directors whose terms expire at the Annual Meeting are Michael Harrington, R. David Hoover, Deborah KochevarWilliam Doyle, Art Garcia, Denise Scots-Knight, and Kirk McDonald.Jeffrey Simmons. Each of these directors has been re-nominated by our Board upon the recommendation of our Corporate Governance Committee (the “Corporate Governance Committee”).Committee. All directors elected at the Annual Meeting will continue in office until the annual meeting of our shareholders to be held in 20262027 and until their successors are elected and qualified.
The four nominees contribute significantly to our Board, including as follows:
| • | All nominees, other than Mr. Simmons, are independent directors; |
| • | Two of the four nominees are public company CEOs; |
| • | One of the four nominees is a former public company CFO; |
| • | One of the four nominees has significant experience leading and serving on public boards of animal health and life sciences companies; and |
| • | Three of the four nominees serve or have served on other public boards. |
Each of the directors nominated by our Board has consented to serving as a nominee for the term listed above, to being named in this Proxy Statement and to serving on our Board for the term listed above, if elected. The persons named as proxies solicited by this Proxy Statement will vote the proxies received by them as directed on the proxy card or, if no direction is made, for the election of each of our Board’s four nominees. If any nominee is unable to serve, our Board can either designate a substitute nominee to serve in his or her place as a director or reduce the size of our Board. If our Board nominates another individual, the persons named as proxies may vote for such substitute nominee. Proxies cannot be voted for a greater number of individuals than the four nominees named in this Proxy Statement.
Our Board has determined that all director nominees, other than Mr. Simmons, are independent of Elanco and management. See “Corporate Governance—Director Independence” below for more information.
Recommendation of the Board
| The Board unanimously recommends a vote “FOR” each of Elanco's Class III director nominees. | ||
| Elanco Animal Health Incorporated | 9 | 2024 Proxy Statement |

| Elanco Animal Health Incorporated | 10 | 2024 Proxy Statement |

| Elanco Animal Health Incorporated | 11 | 2024 Proxy Statement |
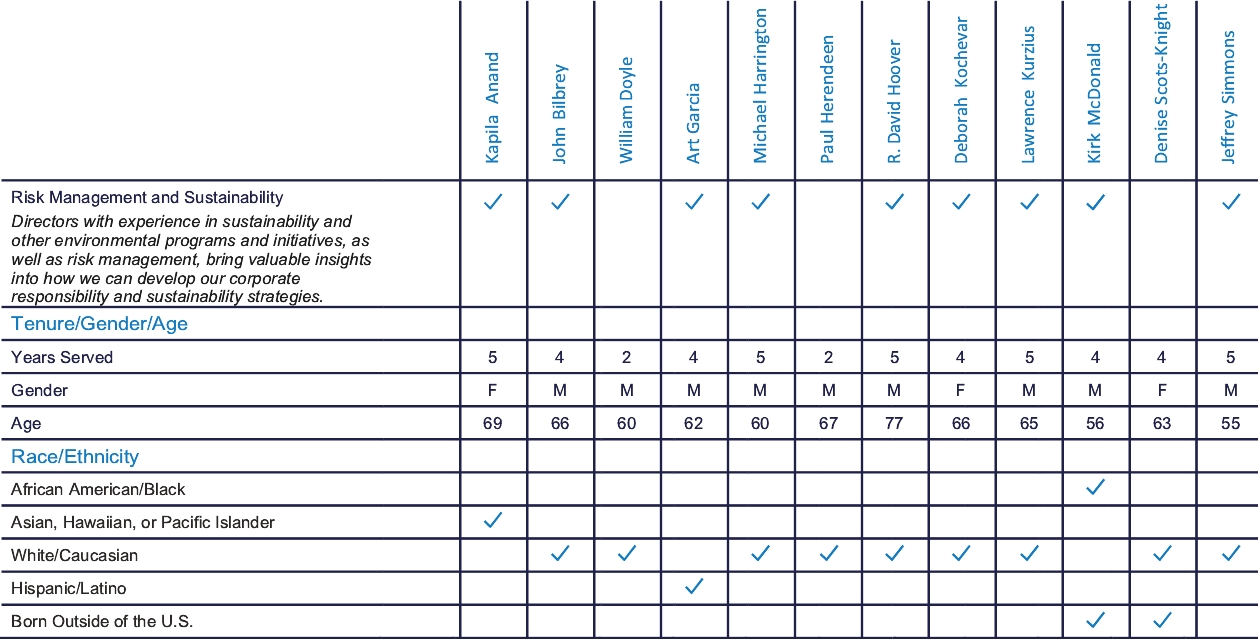
Board Membership Criteria
Our Board is responsible for selecting candidates for Board membership and for establishing the general criteria to be used in identifying potential candidates. The Board has delegated to the Corporate Governance Committee leadsits authority to lead our director succession planning process and regularly considers the criteria necessary to achieve a diverse Board that provides effective oversight of Elanco.
The Corporate Governance Committee believes that all directors should display the personal attributes necessary to be effective directors: integrity, sound judgment, intellectual prowess and versatility, confidence, independence in fact and mindset, ability to operate collaboratively, willingness to ask difficult questions, willingness to listen, the ability to commit the necessary time to duties as a
 |
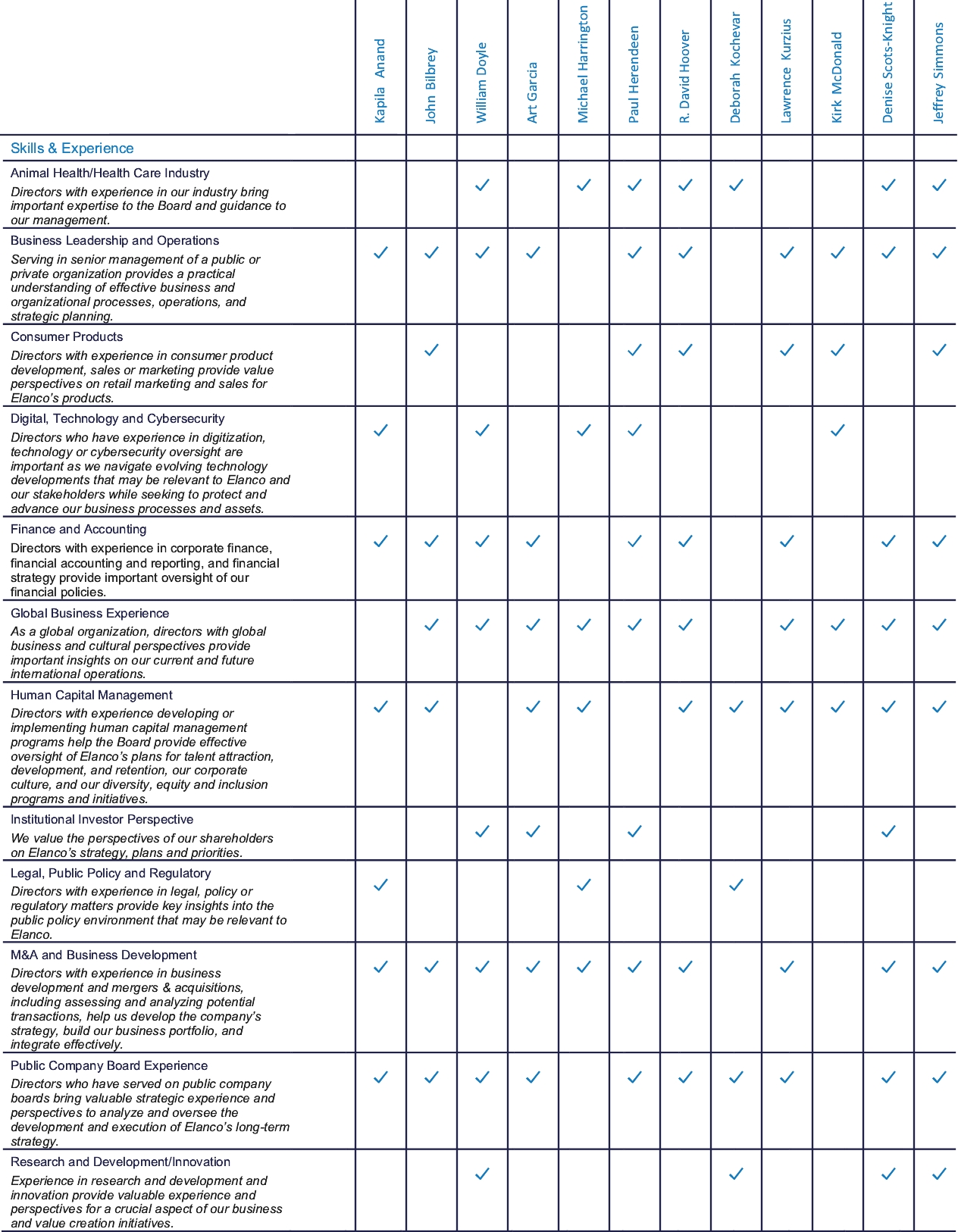
In addition to the above criteria, the Corporate Governance Committee considers, on an ongoing basis, the additional skills, experiences and backgrounds that it seeks in members of our Board in the context of our business and the existing composition of our Board. The directors’ biographies under “—Our Director Nominees and Continuing Directors” below note each director’s relevant skills, experiences and backgrounds that make them suited to contribute to our Board.
Public Company Board Commitments
Our Corporate Governance Guidelines state that directors shall ensure that existing or future commitments do not materially interfere with their ability to fulfill their responsibilities as Elanco directors, given that serving on our Board requires significant time and attention. In general, directors who are not Elanco employees (“non-employee directors”) may not serve on more than three other public company boards and our Chief Executive Officer may not serve on more than one other public company board.
| 12 | 2024 Proxy Statement |  |
 |
 |
Our Board and the Corporate Governance Committee believe that each of our nominees brings a strong and diverse set of skills, experiences and perspectives that, when combined with the other continuing directors, creates a high-performing Board that is aligned with our business strategy and that contributes to the effective oversight of Elanco. The ages, principal occupations, public directorships held and other information about our nominees and continuing directors are shown below.
|
NOMINEES FOR TERMS EXPIRING IN
 | William Doyle EXPERIENCE ● Executive Chairman, Novocure Ltd., a commercial stage oncology company (since 2016) ● Managing Director, WFD Ventures LLC, a private venture capital firm he co-founded (2002 – 2022) ● Senior Advisor and member of the investment team, Pershing Square Capital Management (2013 – 2016) ● Johnson & Johnson (1995 – 1999) - Member, Medical Devices and Pharmaceutical Group Operating Committee - Vice President, Licensing and Acquisitions - Chairman, Medical Devices Research and Development Council - Worldwide President, Biosense-Webster, Inc. - Member of the boards of Cordis Corporation and Johnson & Johnson Development Corporation, Johnson & Johnson’s venture capital subsidiary ● Management consultant, McKinsey & Company OTHER CURRENT AND PRIOR PUBLIC COMPANY BOARDS ● ProKidney Corp. (since 2022) ● Novocure Ltd. (since 2004); Chairman (2009 – 2016) ● Minerva Neurosciences, Inc. (2017 – 2023) ● OptiNose, Inc. (2004 – 2020) ● Zoetis Inc. (2015 – 2016) KEY QUALIFICATIONS ● Animal Health/Health Care Industry experience, gained through his service in roles of increasing responsibility at Johnson & Johnson, his current role at Novocure, and as a director of companies in the healthcare sector, such as OptiNose and Minerva Neurosciences, and the pet health sector, such as Zoetis ● M&A and Business Development experience acquired through his oversight responsibilities while at Johnson & Johnson's venture capital arm and illustrated by Novocure revenue growth of more than $500 million and adjusted EBITDA growth by hundreds of millions of dollars while he served as its Executive Chairman ● Research and Development/Innovation expertise developed through his co-founding and service as Managing Director of WFD Ventures, a technology and life sciences focused venture capital firm, which resulted in a broad understanding of new technologies and emerging business models and risks, as well as through his tenure at Johnson & Johnson, where he managed innovation programs ● Institutional Investor Perspective gained while serving at Pershing Square, a well-known activist hedge fund | |||
Age: 61 Independent Director since: December 2020 | ||||
BOARD COMMITTEES Finance, Strategy and Oversight Innovation, Science and Technology | ||||
QUALIFICATIONS
| ||||
| Elanco Animal Health Incorporated | 13 | 2024 Proxy Statement |
  | Art Garcia EXPERIENCE ● Ryder System, Inc., a North American provider of transportation and supply chain management products known for its fleet of rental trucks - Executive Vice President and Chief Financial Officer (2010 – 2019) - Senior Vice President and Controller (2005 – 2010) - Vice President and Controller (2002 – 2005) OTHER CURRENT PUBLIC COMPANY BOARDS ● Raymond James & Associates (since 2023) ● American Electric Power Company, Inc. (since 2019) ● ABM Industries Incorporated (since 2017) KEY QUALIFICATIONS ● Business Leadership and Operations expertise acquired through his experience leading the finance organization at Ryder Systems, where he led the re-engineering of the organization to help drive efficiency, established a new business model and implemented strategies to revitalize growth and improve profitability ● Finance and Accounting experience developed during the 18 years he served in financial roles at Ryder Systems, where he ultimately had oversight of the entire financial function for almost a decade and during his service on the audit, risk management and governance committees on the boards of other public companies ● M&A and Business Development expertise obtained while overseeing the corporate strategy and business development functions and managing the financial integration of numerous acquisitions at Ryder Systems ● Institutional Investor Perspective developed through his nearly 10 years of experience engaging with the financial community as a public company Chief Financial Officer | |||
Age: Independent Director since: May 2019 | ||||
BOARD COMMITTEES Audit Finance, Strategy and Oversight | ||||
QUALIFICATIONS
| ||||
| Elanco Animal Health Incorporated | 14 | 2024 Proxy Statement |
 | Denise Scots-Knight EXPERIENCE ● Co-Founder, Chief Executive Officer and Director, Mereo BioPharma Group plc, an international biopharmaceutical company focused on oncology and rare diseases (since 2015) ● Partner, Phase4 Ventures GP Ltd., Phase4 Ventures GP III Ltd., Phase4 Ventures III FPGP Ltd., Phase4 Ventures III GP LP, and Phase4 Ventures III FP LP, privately held global life science venture capital firms (2010 – 2019) ● Chief Executive Officer, Phase4 Partners Ltd., a privately held, global life science venture capital firm (2010 – 2015) ● Head, Nomura Phase4 Ventures, a venture capital affiliate of Nomura International plc, a leading Japanese financial institution (2004 – 2010) OTHER CURRENT AND PRIOR PUBLIC COMPANY BOARDS ● Mereo BioPharma Group plc (since 2015) ● OncoMed Pharmaceuticals Inc. (2008 – 2018) ● Albireo Pharma, Inc. (2008 – 2017) KEY QUALIFICATIONS ● Health Care Industry experience acquired over her career in the life sciences industry, and through her current and past service as a director of other public and privately held biotech and life sciences companies and supported by being named of one of the 15 leading women in European biotech by Labiotech UG ● Global Business Experience gained through her service as Co-Founder and Chief Executive Officer of Mereo BioPharma, a United Kingdom-based, Nasdaq-listed company with operations in the U.S., as well as leadership roles in other non-U.S. organizations, which further developed her valuable insights into global strategic oversight, talent and leadership development that are critical in our growth-oriented industry ● Institutional Investor Perspective obtained through her extensive experience investing and allocating capital as the head of a life sciences-focused venture capital firm ● Research and Development/Innovation expertise developed through her career, where she has a track record of building new innovation models and strategic partnerships for emerging technologies, which has resulted in her having a deep acumen and technical expertise beneficial for overseeing our research and development activities | |||
Age: 64 Independent Director since: March 2019 | ||||
BOARD COMMITTEES Compensation and Human Capital Innovation, Science and Technology | ||||
QUALIFICATIONS
| ||||
| Elanco Animal Health Incorporated | 15 | 2024 Proxy Statement |
 | Jeffrey Simmons EXPERIENCE ● President and Chief Executive Officer, Elanco Animal Health Incorporated (since 2018) ● Eli Lilly and Company, a global pharmaceutical company (1989 - 2018) - Senior Vice President and President of the Elanco Animal Health division at Eli Lilly (2008 – 2018) - Held various leadership roles in the Elanco Animal Health division, including International Marketing Manager, Country Director for Brazil, Area Director for Western Europe and Executive Director for U.S. and Global Research & Development KEY QUALIFICATIONS ● Business Leadership and Operations expertise exhibited as a proven, purpose-driven leader during his 30-plus years in the life sciences industry, including as the head of Elanco for the past decade, during which he directed Elanco's growth and transformation from a primarily U.S. livestock feed additive company to a global animal health leader with a diversified business, more than quadrupled revenue, created a unique innovation engine and built five new businesses, including a greater than $2 billion pet health business ● M&A and Business Development experience acquired while evaluating, executing and integrating several significant acquisitions during his tenure as Elanco’s CEO, including the acquisitions of Aratana Therapeutics, Kindred Biosciences and Bayer Animal Health, one of the largest animal health transaction to date ● Risk Management and Sustainability expertise shown when under his leadership, Elanco deepened its commitment to sustainability and, in October 2020, became the first independent animal health company to launch sustainability commitments connected to the United Nations Sustainable Development Goals; also demonstrated by his role in progressing Bovaer, Elanco’s methane reduction product, and developing a carbon insetting marketplace ● Research and Development/Innovation experience acquired through his oversight of research and development programs over the past three decades, including the successful product launch of numerous animal health blockbuster drugs while serving as Executive Director for U.S. and Global Research & Development as well as other senior leadership roles within the Elanco Animal Health Division of Eli Lilly | |||
Age: 56 Director since: September 2018 | ||||
BOARD COMMITTEES Finance, Strategy and Oversight | ||||
QUALIFICATIONS
| ||||
| Elanco Animal Health Incorporated | 16 | 2024 Proxy Statement |
CLASS I DIRECTORS – TERMS EXPIRING IN 2025
 | Kapila Anand EXPERIENCE ● KPMG LLP, one of the world's leading accounting firms - Senior Advisor (2016 - 2020) - Audit Signing Partner and Advisory Partner (1989 - 2016) - Elected to KPMG's U.S. and Americas boards (2005 - 2010) and Chair of the KPMG Foundation OTHER CURRENT AND PRIOR PUBLIC COMPANY BOARDS ● Omega Healthcare Investors Inc. (since 2018) ● Extended Stay America, Inc. and its REIT subsidiary, ESH Hospitality, Inc. (2016 - 2021) KEY QUALIFICATIONS ● Finance and Accounting experience gained through her more than 30 years of services as a Certified Public Accountant at KPMG and utilized during her service on the audit committees of multiple public companies ● Risk Management and Sustainability expertise developed over her career advising companies throughout their life cycle on topics such as strategic planning, due diligence, risk assessments, enterprise risk management, and setting up proper internal controls and further honed through her work as an advisory partner to KPMG's risk and governance practice ● Legal, Public Policy and Regulatory experience acquired while playing a leading role in the development of KPMG's private equity and regulatory businesses ● M&A and Business Development experience obtained while acting as an advisory partner to KPMG's M&A and integration services practie | |||
Age: 70 Independent Director since: September 2018 | ||||
BOARD COMMITTEES Audit (Chair) Corporate Governance | ||||
QUALIFICATIONS
| ||||
| Elanco Animal Health Incorporated | 17 | 2024 Proxy Statement |
 | John (JP) Bilbrey EXPERIENCE • Executive Chair, Olaplex Holdings, Inc., a science-enabled beauty company (since 2023) • Advisor, Royal Bank of Canada, a multinational financial services company (2019 – 2020) • The Hershey Company, a global consumer food company (2003 - 2017) - Chairman, Chief Executive Officer and President (2015 – 2018) - Chief Executive Officer and President (2011 – 2017) - Held roles of increasing responsibility including EVP, Chief Operating Officer; President of North America; and President of the International Commercial group • Held leadership positions at Mission Foods; Danone Waters of North America, Inc.; Bilbrey Farms and Ranch; and The Procter & Gamble Company OTHER CURRENT AND PRIOR PUBLIC COMPANY BOARDS • Olaplex Holdings, Inc. (since 2023) • Tapestry, Inc. (since 2020) • Colgate-Palmolive Company (since 2015) • Campbell Soup Company (2019 – 2023) • The Hershey Company (2011 - 2018); Chairman (2015 - 2018) • McCormick & Company, Inc. (2005 – 2015) KEY QUALIFICATIONS • Consumer Products experience and deep knowledge gained over his long history of successfully building and marketing brands in the consumer products industry, including 15 years of leadership experience at Hershey and 22 years at Procter & Gamble • M&A and Business Development expertise demonstrated by his track record of successfully buying and integrating companies and growing and leading businesses in the consumer products industry • Finance and Accounting expertise developed as a Certified Public Accountant and deepened while overseeing Hershey’s financial and accounting practices, operating budgets and financial statements, as Chairman and Chief Executive Officer of a global food products leader • Unique combination of livestock production, food industry and consumer insights experience, all of which are highly relevant to our industry, due to service as an owner and operator of commercial cattle operations for Bilbrey Farms and Ranch | |||
Age: 67 Independent Director since: March 2019 | ||||
BOARD COMMITTEES Audit Finance, Strategy and Oversight (Chair) | ||||
QUALIFICATIONS
| ||||
| Elanco Animal Health Incorporated | 18 | 2024 Proxy Statement |
 | Paul Herendeen EXPERIENCE • Bausch Health Companies Inc., a global health care products company - Advisor to the Chairman and Chief Executive Officer (2021 – 2022) - Executive Vice President and Chief Financial Officer (2016 – 2021) • Executive Vice President and Chief Financial Officer, Zoetis Inc., an animal health company (2014 – 2016) • Chief Financial Officer, Warner Chilcott, a specialty pharmaceuticals company (2005 – 2013; 1998 – 2001) • Executive Vice President and Chief Financial Officer, MedPointe Pharmaceuticals, a privately held pharmaceutical company • Principal investor, Dominion Income Management, the predecessor to Cascade Investment, which managed investments on behalf Cornerstone Partners • Held various positions with the investment banking group of Oppenheimer & Company and the capital markets group of Continental Bank Corporation • Senior auditor, Arthur Andersen & Company KEY QUALIFICATIONS • Animal Health/Health Care Industry experience gained serving in leadership positions at MedPointe Pharmaceuticals, Warner Chicott, Zoetis, and Bausch Health over more than 20 years • Finance and Accounting expertise developed through decades of experience serving in financial roles in the life sciences industry, including service as the Chief Financial Officer of Zoetis, and at Bausch Health, where he helped the company reduce its debt and strengthen its balance sheet • M&A and Business Development experience from his tenure at Warner Chilcott, MedPointe Pharmaceuticals, Zoetis and Bausch Health, as well as his nearly decade of experience as a principal at Dominion Income Management and Cornerstone Partners, where he worked on investments as well as mergers and acquisitions for the firms and their portfolio companies • Institutional Investor Perspective developed through his more than 15 years of experience engaging with the financial community as a public company Chief Financial Officer and leader of award-winning investor relations programs | |||
Age: 68 Independent Director Since: December 2020 | ||||
BOARD COMMITTEES Audit Finance, Strategy and Oversight | ||||
QUALIFICATIONS
| ||||
| Elanco Animal Health Incorporated | 19 | 2024 Proxy Statement |
 | Lawrence Kurzius EXPERIENCE • McCormick & Company, Inc., a global Fortune 500 food company (since 2005) - Executive Chairman (since 2023) - Chairman and Chief Executive Officer (2017 - 2023) - Chief Executive Officer (2016) - Held various leadership roles, including President and Chief Operating Officer; Chief Administrative Officer; President, International Businesses; President, Europe, Middle East and Africa; President, U.S. Consumer Foods • Zatarain's, an American food company - President and CEO (1997 - 2004 when acquired by McCormick) - Vice President and Marketing Director (1991 - 1997) • Marketing executive, Quaker Oats Company and Mars Inc.'s Uncle Ben's Company OTHER CURRENT PUBLIC COMPANY BOARDS • The Cooper Companies, Inc. (since 2023) • McCormick & Company, Inc. (since 2015), Executive Chairman (since 2023), Chairman (2017 - 2023) KEY QUALIFICATIONS • Consumer Products experience acquired over his career in consumer goods marketing and senior leadership roles at Mars, Quaker Oats, Zatarain's and McCormick, a large, multi-faceted, consumer and flavor solutions food business, and further developed while serving on the boards of multiple industry groups, including The Consumer Goods Forum, The Consumer Brands Association and The National Association of Manufacturers, resulting in his extensive knowledge of consumer trends and a deep understanding of consumer preferences • Global Business Experience gained leading multinational companies, where understanding both the domestic and international markets was essential to success • Human Capital Management expertise developed through his leadership of a company with over 14,000 employees globally, which has resulted in a deep understanding of attracting, developing, motivating and retaining top talent, as well as executive compensation and leadership development • Risk Management and Sustainability experience obtained from his broad executive experience at McCormick, where under his leadership, the company became a UN Global Compact LEAD company while embedding purpose-led performance into McCormick's culture by championing the company's industry-leading sustainability efforts | |||
Incoming Chairman Age: 66 Independent Director Since: September | ||||
BOARD COMMITTEES Compensation and Human Capital (Chair) Corporate Governance | ||||
QUALIFICATIONS
| ||||
| Elanco Animal Health Incorporated | 20 | 2024 Proxy Statement |
 | Craig Wallace EXPERIENCE • President, C.S. Wallace Investments + Strategy, a firm that invests and advises early to mid-stage companies in agribusiness, animal health and human healthcare (since 2019) • Chief Executive Officer, Hannah Pet Hospitals, a veterinary hospital and animal care concept based in Portland, Oregon (2019 – 2021) • Chief Executive Officer and North America Pacific Zone Director, Ceva Santé Animale, a multinational animal health company (2011 – 2019) • Senior Vice President Sales and Marketing, Trupanion, Inc., a pet insurance provider (2010 – 2011) • Fort Dodge Animal Health, a leading global manufacturer of animal health products for the livestock, companion animal, equine, swine, and poultry industries (1989 – 2009) - Held roles of increasing responsibility, including Vice President of U.S. Sales and Vice President of U.S. Marketing KEY QUALIFICATIONS • Animal Health Industry experience gained through his nearly 35 years of service in animal health • Business Leadership and Operations experience gained as | • Consumer Products experience through his more than two decades at Fort Dodge Animal Health and Ceva Santé Animale, global manufacturers of animal health products • Institutional Investor Perspective developed through his experience leading an investment and advisory firm that engages with companies in our industry | |||
Age: 60 Independent Director since: March 2024 | |||||
BOARD COMMITTEES Finance, Strategy and Oversight | |||||
QUALIFICATIONS
| |||||
| Elanco Animal Health Incorporated | 21 | 2024 Proxy Statement |
CLASS II DIRECTORS – TERMS EXPIRING IN 2026
 | Michael Harrington EXPERIENCE •Eli Lilly and Company, a global pharmaceutical company (1991 - 2020) - Senior Vice President, General Counsel - Vice President and Deputy General Counsel, - Vice President and Deputy General Counsel, Corporate (2004 – 2010) - Managing Director of
| |||
- General Counsel, Asia Pacific (1996 - 2000) OTHER CURRENT PUBLIC COMPANY BOARDS
• KEY QUALIFICATIONS • Animal Health/Health Care Industry experience in more than three decades at Eli Lilly, one of the world’s leading global pharmaceutical companies and • Digital, Technology and Cybersecurity expertise • Legal, Public Policy and Regulatory expertise developed and demonstrated having responsibility and oversight of legal and public policy issues, government and regulatory affairs, intellectual property, risk management, corporate governance and compliance
• | ||||
Age: 61 Independent Director Since: September 2018 | ||||
BOARD COMMITTEES Innovation, Science and Technology | ||||
QUALIFICATIONS
| ||||
| 22 | 2024 Proxy Statement |  |
  | | |||
R. David Hoover | ||||
EXPERIENCE
• - Chairman (2002 – 2011) - Chief Executive Officer (2001 – 2011) - President (2000 – 2010) - Chief Operating Officer (2000 – 2001) - Vice Chairman and Chief Financial Officer
| ||||
OTHER PRIOR PUBLIC COMPANY BOARDS • Edgewell Personal Care,
• Eli Lilly and Company (2009 - 2018) • Ball Corporation (1996 – 2018)
| ||||
• Steelcase Inc. (2016 – 2016) KEY QUALIFICATIONS
• • Consumer Products expertise gained
• • M&A and Business Development experience gained while at Ball, where he was instrumental as the chief strategist and lead negotiator for Ball in the largest acquisition in the company's history, the purchase of the | ||||
Chairman of the Board Age: 78 Independent Director Since: September 2018 | ||||
BOARD COMMITTEES Compensation and
Corporate Governance (Chair) | ||||
QUALIFICATIONS
| ||||
| 23 | 2024 Proxy Statement |  |
  | | |||
Deborah Kochevar, DVM, Ph.D, DACVCP | ||||
EXPERIENCE
•Tufts University, a private research university - Senior Fellow, The Fletcher School of Law and Diplomacy and Dean Emerita, Cummings School of Veterinary - Provost and Senior Vice President ad interim (2018 – 2019) - Dean of the Cummings School of Veterinary Medicine (2006 – 2018)
• Long-time faculty member and administrator, College of Veterinary Medicine and Biomedical Sciences at Texas A&M University, held the Wiley Chair of Veterinary Medical Education
• Boarded diplomate of the American College of Veterinary Clinical Pharmacology
| ||||
OTHER CURRENT PUBLIC COMPANY BOARDS • Charles River Laboratories International, Inc.
| ||||
KEY QUALIFICATIONS •
• Legal, Public
• • Risk Management and Sustainability knowledge acquired from her | ||||
Age: 67 Independent Director Since: March 2019 | ||||
BOARD COMMITTEES Corporate Governance Innovation, Science and | ||||
QUALIFICATIONS
| ||||
| 24 | 2024 Proxy Statement |  |
  | | |||
Kirk McDonald | ||||
EXPERIENCE
• Chief Executive Officer and member of Global Leadership Team, GroupM North America, a global provider of media and advertising solutions through the development of technology-enabled services
•Xandr, Inc., AT&T's advertising division - Chief Business Officer - Chief Marketing Officer
• - Chief Marketing Officer, Advertising and Analytics, AT&T Inc. - President, - President, - Chief Advertising Officer, Fortune|Money - Senior Vice President of Network Sales, DRIVEpm and Atlas (both units of Microsoft's advertising business)
| ||||
OTHER CURRENT PUBLIC COMPANY BOARDS • Ziff Davis, Inc. (since 2023)
KEY QUALIFICATIONS • • Consumer Products experience gained during his more than 30 years of experience in marketing • Digital, Technology and Cybersecurity expertise developed at GroupM, where he helped develop technology-enabled services to provide media and advertising solutions, resulting in experience with digital and emerging
• Human Capital Management insights gained as Chief Executive Officer of GroupM, an organization of approximately 6,500 people in a fast-growing industry | ||||
 |
 | |||||
Age: Independent Director Since: March 2019 | |||||
BOARD COMMITTEES Compensation and Innovation, Science and Technology | |||||
| |||||
| 25 | 2024 Proxy Statement |  |
 | |||||
 | |
Kathy Turner
EXPERIENCE • - Senior Vice President and Chief - Corporate Vice President, - Corporate Vice President,
| |
• | |
 |
 | |
| |
 |
 | |
 |
 | |
 |
 | |
| |
KEY QUALIFICATIONS •
| |
• Business Leadership and Operations experience gained through her positions of increasing responsibility at IDEXX Laboratories and Abbott Laboratories • |
 |
 | |
 |
 | ||||
• Consumer Products experience gained through 10 years of strategy development and commercial experience in multiple roles at Abbott Laboratories in the Nutritional Products Division and Diabetes Care Division • M&A and Business Development experience gained through her position as Divisional Vice President, Global Strategic Operations, Abbott Diagnostics Division of Abbott Laboratories • Research and Development/Innovation experience gained through her position as Divisional Vice President, Global Strategic Operations, Abbott Diagnostics Division of Abbott Laboratories | ||||
Age: 60 Independent Director since: March 2024 | ||||
BOARD COMMITTEES Finance, Strategy and Oversight | ||||
QUALIFICATIONS
| ||||
| 26 | 2024 Proxy Statement |  |
 | ||
Corporate Governance | ||
 |
We are committed to the values of effective corporate governance and high ethical standards. Westandards, which we believe these values are conducive tosupport long-term performance. We believe ourOur key corporate governance and ethics policies help enable us to manage our business in accordance with the highest standards of business practice and in the best interests of our shareholders.
Our Corporate Governance Guidelines and committee charters help govern the operation of our Board and its committees in executing their responsibilities. These are reviewed at least annually by the Corporate Governance Committee and the Board and are updated periodically in response to changing regulatory requirements, evolving practices, issues raised by our shareholders and other stakeholders and otherwise as circumstances warrant.
Recent Corporate Governance Enhancements
Our Board regularly evaluates our governance-related practices,the Company’s governance profile, taking into account evolving best practices,standards, the needs of our business and feedback we receive from our shareholders and other stakeholders, including as described in “—Shareholder Outreach”“Shareholder Engagement” below. InWe have engaged extensively with our shareholders over the last three years on corporate governance matters, and in 2022, due in partwe began a process to evolve the Company’s governance practices in response to thisshareholder feedback. Last year, we eliminated the supermajority vote requirements to amend the Company’s Articles of Incorporation and Bylaws. We also adopted proxy access.
As part of a comprehensive governance review following the Company’s 2023 Annual Meeting, and in response to shareholder feedback, we have madein the following enhancements:fall of 2023, the Board initiated a process to implement several additional governance enhancements, including:
| ● | Submitting for shareholder approval at this annual meeting amendments to the Company’s Articles of Incorporation to: |
| ◦ |
| ◦ |
| ◦ |
| ◦ | Allow shareholders under certain circumstances to request special meetings of shareholders |
| ● | Implementing a |
| ● | Rotating Board committee leadership, with Michael Harrington assuming the role of | |||||||
| ● | Enhancing disclosure in our annual ESG report and | |||||||
|  |
We believe these enhancements demonstrate our responsiveness to shareholder feedback and commitment to goodeffective corporate governance. Wegovernance and ouroversight. Our Board areis committed to continuing to drive progress regarding our corporate governance and looklooks forward to continuing our dialogue with our shareholders and other stakeholders on these topics.
We also recently appointed two new directors to our Board: Kathy Turner and Craig Wallace. Ms. Turner and Mr. Wallace were identified as potential directors by our shareholder Ancora Catalyst Institutional, LP and certain of its affiliates (together, "Ancora") and were appointed to our Board in accordance with our cooperation agreement with Ancora.
| Elanco Animal Health Incorporated | 27 | 2024 Proxy Statement |
Board Leadership Structure
We have separate roles for the Board Chairman and Chief Executive Officer. As described in our Corporate Governance Guidelines, our Board currently has a strong, independent, non-executive chairman, R. David Hoover, which we believe helps further strengthen our governance structure. OurAt the conclusion of this year’s Annual Meeting, Lawrence Kurzius, an experienced Executive Chairman and Chief Executive Officer and board leader, will succeed Mr. Hoover as Board Chairman.
At this time, our Board believes this structurethe separation of the Board Chairman and Chief Executive Officer roles provides an effective leadership model for Elanco and our Board to help assureensure effective independent oversight at this time.oversight. However, our Board also believes that no single leadership model is right for all companies and at all times. Depending on the circumstances, other leadership models, such as combining the roles of the Board Chairman and Chief Executive Officer, might be appropriate. Our Board periodically reviews its leadership structure and will continue to evaluate and implement the leadership structure that it determines most effectively supports our Board in fulfilling its responsibilities.
In addition to the leadership of our Board Chairman, our independent directors have ample opportunity to and regularly do, assess the performance of our Chief Executive Officer and provide him meaningful direction to him.direction. Our Board has strong and effective independent oversight of management:
| ● |
| ● | ||||
Each member of the Audit Committee, the Corporate Governance Committee, the Compensation and Human Capital Committee, and the Innovation, Science and Technology Committee is independent; |
| ● | ||||
Each chair of our Board’s five standing committees is independent; |
| ● | ||||
Board and committee agendas are prepared by their independent chairs, based on discussions with all directors and recommendations from senior management; and |
| ● | |||||
All directors are encouraged to request agenda items, additional information and/or modifications to schedules as they deem appropriate. |
| Elanco Animal Health Incorporated | 28 | 2024 Proxy Statement |
Board Oversight
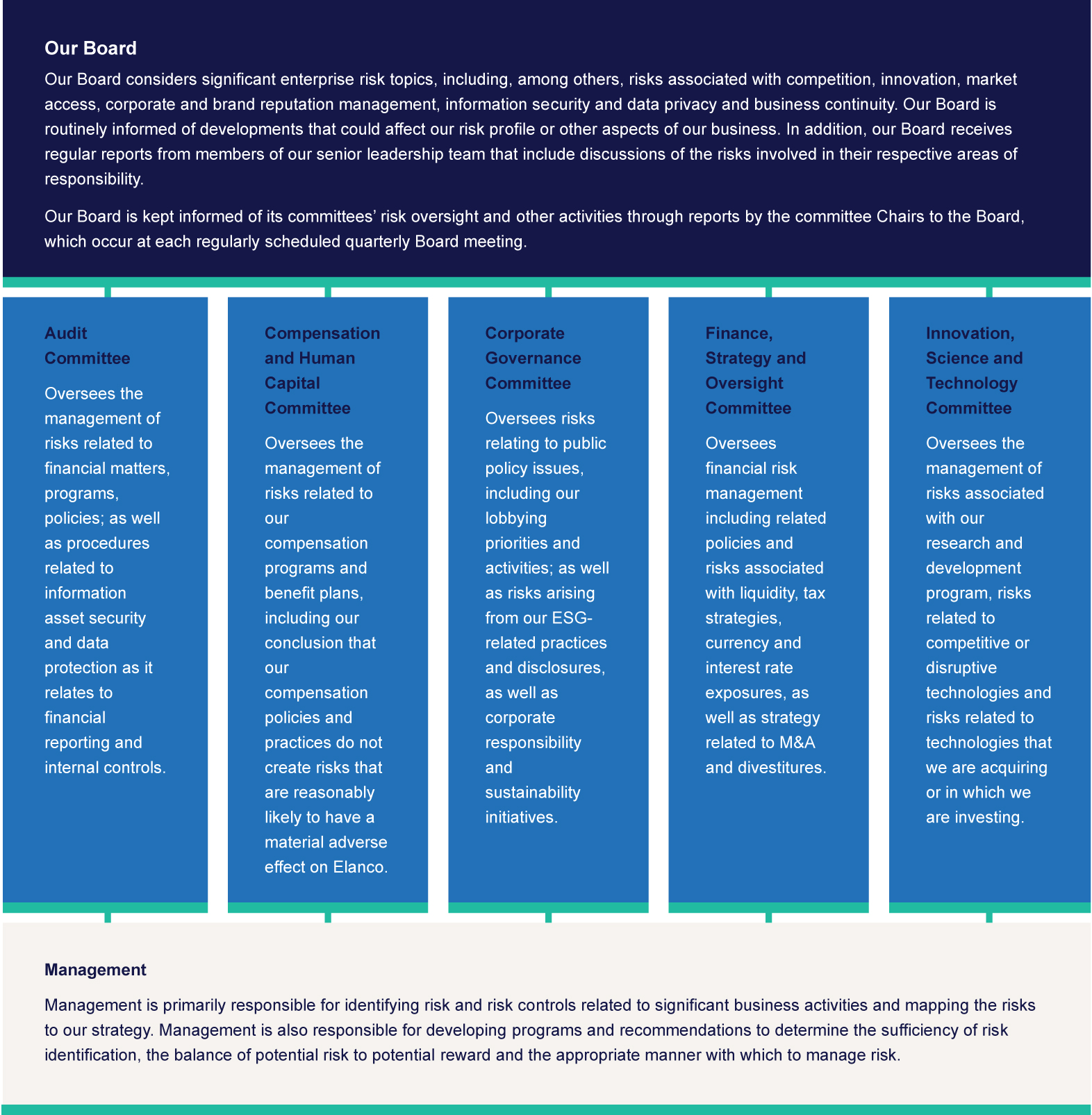
OUR BOARD’S OVERSIGHT OF RISK MANAGEMENT
We have an enterprise risk management program overseen by our Chief Compliance Officer,General Counsel, who is supported by our internal General Auditor. Material enterprise risks, which include competitive, strategic, operational, financial, legal, regulatory and ESG risks, are identified and prioritized by management through both top-down and bottom-up processes. Our management is charged with managing these risks through robust internal processes and controls.
 |
Our Board has responsibility for oversight of our management’s planning for material risks. Our enterprise risk management program is reviewed annually at a Board meeting and enterprise risks are also addressed in periodic business function reviews. Reviews of certain risk areas are also conducted by relevant Board committees, as described below.
 |
OUR BOARD’SBOARD'S OVERSIGHT OF STRATEGY
Our Board and its committees are involved in overseeing our corporate strategy, including major business, organizational and transformational initiatives; capital allocation priorities; and significant acquisitions and other transactions, as well as related integration issues. Our Board engages in robust discussions regarding our corporate strategy at nearly every Board meeting. Our Board’s committees oversee elements of our strategy associated with their respective areas of responsibility.
OUR BOARD’SBOARD'S OVERSIGHT OF HUMAN CAPITAL AND SUCCESSION PLANNING
Our approximately 9,000 full-time global employees help shape the Elanco culture and everything we do for our customers. The Elanco Employee Promise states that together we foster an inclusive culture where everyone can make a difference, encouraging ownership, growth and well-being, while focusing on customers and the animals in their care.
Our Board’s committees oversee elements of our culture associated with their area of responsibility. The Compensation and Human Capital Committee is kept informed of our compensation practices, including pay equity, through recurring updates. The Compensationupdates and Human Capital Committee is responsible for periodically discussing with our management and evaluating our performance in the development, implementation and effectiveness of our policies and strategies related to human capital management and diversity in our workforce. The Audit Committee is responsible for oversight of our ethics and compliance program and regularly receives updates on our culture of integrity and the tone set by leaders throughout the organization.
Succession planning for our senior leadership positions is critical to our success. The Compensation and Human Capital Committee reports to our Board on succession planning and leadership development for our Chief Executive Officer as well as certain other executive positions. This topic is discussed formally at least once per year and is also discussed regularly in executive session. The Corporate Governance Committee is tasked with focusing on director succession planning. In performing this function, the committee is responsible for recruiting and identifying nominees for election as directors to our Board.
OUR BOARD’SBOARD'S OVERSIGHT OF INFORMATION SYSTEMS AND CYBERSECURITY
We prioritize the trust and confidence of our customers and workforce.workforce, and we evaluate cybersecurity risks on an ongoing basis. Both management and our Board have an overall responsibility for assessing and managing risks from cybersecurity threats. Our dedicated Chief Information Security Officer (“CISO”) is responsible for leading an information security team that helps prevent, identify and appropriately address cybersecurity threats. The team focuses on developingis responsible for the design and implementing strategies and processes to protect the confidentiality, integrity and availabilityexecution of our assets.
We have been buildingbuilt a risk-based, fit-for-purpose and innovative information security program. Our information security architecture is designed to acceptfocused on designing IT-related solutions that are foundationally secure which accepts and embraceembraces the realities of modern working with a modern, cloud heavy footprint and extended remote workforce. Overall, our program leveragesapproach to cybersecurity governance, risk and aligns with various frameworkscompliance is based on overarching guidelines, standards and goodbest practices includingdeveloped by the U.S. National Institute of Standards and Technology (NIST) Cyber Security Framework, ISO 27000 family(“NIST”), a department of Standards, Information Technology Infrastructure Library (ITIL) Processes and other good practice control methods.the U.S. Department of Commerce. We expect to continue to mature and enhance our information security program as we progress.program.
The Board oversees our cybersecurity management processes, including appropriate risk mitigation strategies, systems, processes and controls. Our CISO meets with the Audit Committee oversees our program,and separately with the full Board at least twice annually to discuss the status of policies and procedures related to information assetsecurity. These discussions focus on any notable incidents and incident responses, updates on known or perceived cyber threats and the information security team's recent actions taken in response to any such incidents and data protection as it relates to financial reporting and internal controls, including data privacy and network security. Broad oversight is maintained by our Board. Thethreats. In addition, the Audit Committee and ourthe Board regularly receive reportsupdates from the CISO and/or our Chief Information Security Officer on, among other things, assessments of risks and threats to our security systems and processes to maintain and strengthen information security systems. Our Chief Information Security Officer also meets with the Audit Committee at least annually in executive session without other members of our management present.as needed.
| Elanco Animal Health Incorporated | 30 | 2024 Proxy Statement |
OUR BOARD’SBOARD'S OVERSIGHT OF ESG AND SUSTAINABILITY
Our Board and executive management team have identified the management of ESG issues and related sustainability efforts as an important priority for Elanco. Leadership across the company actively guides these efforts, including our management Executive Committee, multiple steering committees and issue-specific working groups led by senior managers and employees across our business.
 |
The Global Head of ESG and Sustainability provides quarterly updates about our ESG and sustainability program to the Corporate Governance Committee, including an annual update to the full Board.
| Elanco Animal Health Incorporated | 31 | 2024 Proxy Statement |
Board and Committee Information
During 2022,2023, our Board met sixeight times. Each of our directors other than Mr. Doyle, attended at least 75% of the total meetings of our Board and the Board committees on which he or she served. As noted in his biography on page 14, Mr. Doyle possesses a unique set of skills and experiences that greatly benefit the Board and the committees on which he serves, including his prior service on the Board of Zoetis as well as other public and private companies, his leadership through and after the initial public offering of Novocure and his understanding of and experience with new technologies. Mr. Doyle has assured the Company of his commitment to serving on the Board and his intention to attend a greater number of meetings in 2023. Consistent with the expectations in our Corporate Governance Guidelines, each of our directors attended our 20222023 annual meeting of shareholders.
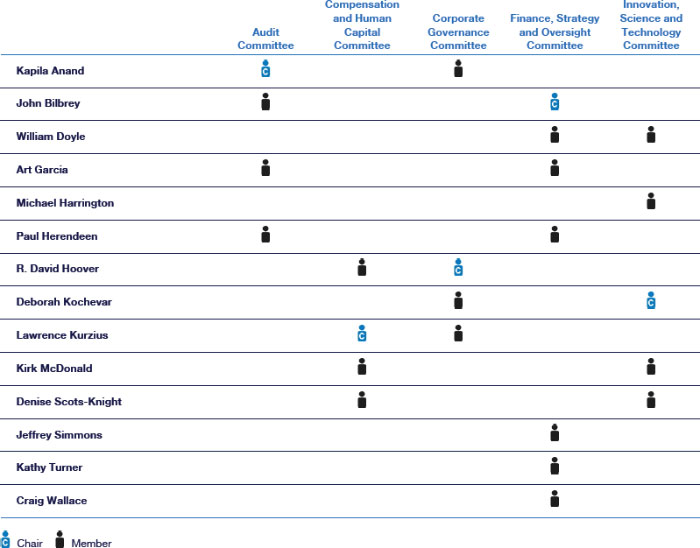
The table below provides the current membership of each of the standing Board committees.
| 32 | |||||||||||||||||
 |  | ||||||||||||||||
 |  | ||||||||||||||||
 |  | ||||||||||||||||
 |  | ||||||||||||||||
 | |||||||||||||||||
 |  | ||||||||||||||||
 |  | ||||||||||||||||
 |  | ||||||||||||||||
 |  | ||||||||||||||||
 |  | ||||||||||||||||
 |  | ||||||||||||||||
 |


 |
BOARD COMMITTEES
Audit Committee
Membership: Kapila Anand (Chair) Art Garcia John Bilbrey Paul Herendeen Meetings in 2023: 11 | Key Assist our Board in its oversight of:
• the integrity of our financial statements and any other financial information that is provided to our
• the independent auditor’s qualifications and independence;
• the systems of internal controls and disclosure controls that our management has established;
• the performance of our internal and independent audit functions;
•our compliance with legal and regulatory
• information security and data privacy matters as they relate to financial reporting and internal controls. The Audit Committee is also directly responsible for the appointment, compensation, retention and | |||||
| Our Board has determined that each member of the Audit Committee is independent within the meaning of our independence standards and applicable rules and regulations of the U.S. Securities and Exchange Commission (the “SEC”) and applicable NYSE listing standards. Each member is also financially literate, and Ms. Anand qualifies as an “audit committee financial expert.” | ||||||
| 33 | 2024 Proxy Statement |  |
Compensation and Human Capital Committee
Membership: Lawrence Kurzius (Chair)
R. David Hoover Denise Scots-Knight Kirk McDonald |
Meetings in 2023: 5 | Key Responsibilities: Assist our Board in its oversight • our
• Elanco's performance in the area of diversity in its workforce; and
Each Compensation and Human Capital Committee member is a “non-employee director” as defined in Rule 16b-3 under the Securities Exchange Act of 1934, as amended (the “Exchange Act”). The Compensation and Human Capital Committee’s charter is available on our website at www.elanco.com/en-us/about-us/governance/corporate by clicking on the “Compensation and Human Capital Committee Charter” link. Compensation Committee Interlocks and Insider Participation: During | | ||||
| Our Board has determined that each member of the Compensation and Human Capital Committee is independent within the meaning of our independence standards and applicable NYSE listing standards. | |||||||
| Elanco Animal Health Incorporated | 34 | 2024 Proxy Statement |
Corporate Governance Committee
Membership: R. David Hoover (Chair) Kapila Anand Deborah Kochevar Lawrence Kurzius Meetings in 2023: 5 | Key Assist our Board in its oversight of:
• recommending qualifications required for membership on our Board and its committees;
• identifying and recommending candidates for membership on our Board and its committees;
• developing and recommending criteria and policies relating to the services of directors;
• risk management related to
• current and emerging political, social and environmental trends and public policy issues that may affect
The Corporate Governance Committee’s charter is available on our website at | |||||
| Our Board has determined that each member of the Corporate Governance Committee is independent within the meaning of our independence standards. | ||||||
 |
Finance, Strategy and Oversight Committee
Membership: John Bilbrey (Chair) William Doyle Art Garcia Paul Herendeen Jeffrey Simmons Kathy Turner Craig Wallace Meetings in 2023: 4 | Key Assist our Board in its oversight of:
• certain of our financial policies, plans and transactions, including mergers and acquisitions (including
• financial risk management, including oversight of Elanco’s financial risk management policies, and risks associated with liquidity, tax strategies, currency and interest rate exposures; and • our progress The Finance, Strategy and Oversight Committee’s charter is available on our website at www.elanco.com/en-us/ | |||||
| Elanco Animal Health Incorporated | 35 | 2024 Proxy Statement |
Innovation, Science and Technology Committee
Membership: Deborah Kochevar (Chair) William Doyle Michael Harrington Kirk McDonald Denise Scots-Knight Meetings in 2023: 4 | Key Assist our Board in its oversight of:
• our strategy, activities, results and investment in and optimization of research, development, go-to-market strategies and supporting investments, external innovation/business development and innovation initiatives;
• our strategic, tactical and policy matters related to science and technology and any changes to the development and regulatory landscape;
• advancement and augmentation of our product pipeline • management of risks related to our research and development program, competitive or disruptive technologies and technologies which Elanco is acquiring or in which we are investing; and our ambition to achieve scientific innovation leadership in the animal health industry. The Innovation, Science and Technology Committee’s charter is available on our website at www.elanco.com/en-us/about-us/governance/corporate by clicking on the “Innovation, Science and Technology Committee Charter” link. | |||||
| Our Board has determined that each member of the Innovation, Science and Technology Committee is independent within the meaning of our independence standards. | ||||||
| 36 | 2024 Proxy Statement |  |
Shareholder OutreachEngagement
We are engagedvalue shareholder feedback and regularly engage in active discussions with our shareholders to facilitate investor understanding aroundgather their feedback on a broad range of subjects, such astopics, including strategy initiatives, business performance, corporate governance, risk and compensation practices and ESGESG-related initiatives and metrics. We believe this approach to engagement drives increased corporate accountability, improves our decision making and ultimately helps create long-term value. We pursue multiple avenues for shareholder engagement, including periodic reports on our activities, in person and virtual meetings with our shareholders around quarterly earnings, attendance at numerous investor and issuingindustry conferences, and periodic reports on our activities. During 2022, we continued our extensive outreach efforts through an integrated group featuring our President and CEO; Chief Financial Officer; EVP, General Counsel and Corporate Secretary; Headgovernance-focused discussions with many of Investor Relations; EVP, U.S. Pet Health and Global Digital Transformation; EVP, Innovation, Regulatory, and Business Development and other commercial leaders.largest shareholders.
In addition to our regular outreachinvestor relations efforts throughout the year, in the fourth quarter of 20222023 and into 2023,2024, we held a series of meetings with many ofundertook our institutional shareholdersannual shareholder engagement initiative focused specifically focused on ESG performancecorporate governance, sustainability and disclosure. As part of this process, we initiated outreachreached out to and engaged with investorsour largest institutional shareholders representing an aggregate of approximately 60% of our outstanding shares. A numbershares (based on ownership reports as of June 30, 2023). Shareholders representing approximately 50% of our directors, includingoutstanding shares (8 of our top 15 shareholders) accepted our invitation to engage and met with us to share their feedback.
These meetings were led primarily by either R. David Hoover, the current Chairman of ourthe Board, engaged with investors as partor Lawrence Kurzius, the incoming Chairman of these discussions. Throughthe Board, and generally included one or more additional independent directors. During these meetings, we discussed and receivedinvited shareholder input and addressed questions on our governance practices, previous low votes received for directors, corporate strategy, and our executive compensation program and governance practices. These engagement efforts allowed us to better understand our shareholders’ priorities and perspectives and provided us with useful input concerningprogram.
Shareholder feedback we received during these topics.
 |
 |
Our approach toBoard considers the management of ESG issues and related sustainability efforts an important priority for Elanco. The Board allocates oversight of ESG and sustainability – called Elanco’s Healthy Purpose™ – is a framework of commitments and actions built on four interconnected pillars: Healthier Animals, Healthier People, Healthier Planet and Healthier Enterprise – which representmatters among its committees as follows:
Corporate Governance Committee
Assists the areas we believe are most important to our customers, employees, investors and other stakeholders.
Compensation and Human Capital Committee oversee Elanco’s managerial approach
Assists the Board in oversight of policies and strategies related to select ESG and sustainability issues. See “—Board Oversight” above.
 |
Audit Committee reviews
Assists the Board in oversight of policies and practices related to environmental protection, monitors compliance and reports or makes recommendations to the Board, as appropriate.
 |
Innovation, Science and Technology Committee oversees our strategy, initiatives and investments
Assists the Board in oversight of ESG issues related to research, development and innovation. The Antimicrobial Policy Teaminnovation initiatives, including changes to the development and Sustainability and ESG Committee provide counsel and execution of strategy. Learn more about Elanco’s antimicrobial stewardship efforts at: https://www.elanco.com/en-us/research-and-development/antibiotic-stewardship.regulatory landscape.
| 37 | 2024 Proxy Statement |  |
Director Independence
Our Board has established that, for a director to qualify as independent, a director must have no material relationship with Elanco other than as a director or, either directly or indirectly, as a partner, significant shareholder or officer of an organization that has a material relationship with Elanco. In making this determination, our Board considers all relevant facts and circumstances and has adopted the categorical independence standards for directors established in the NYSE listing standards.
The Corporate Governance Committee has reviewed the applicable legal and NYSE listing standards for Board and committee member independence. A summary of the responses to annual questionnaires completed by each of the directors and a report of transactions with director-affiliated entities are madeprovided to the committee. On the basis of this review, the committee delivers a recommendation to our Board, which then makes its independence determination.determines independence.
Our Board has determined that each of our directors, other than Mr. Simmons, is independent of Elanco and its management. Mr. Simmons is not independent because he serves as our President and CEO.
In making these determinations, our Board considered that in the ordinary course of business, relationships and transactions may occur between Elanco and our subsidiaries, on the one hand andsubsidiaries. Additionally, entities affiliated with directors or their family members on the other hand.were also considered. Dr. Kochevar is employed at an academic institution and Mr. Garcia’s domestic partner is affiliated with a law firm and Mr. McDonald is employed by a for-profit company with which we have had relationships or transactions in the ordinary course of business. We reviewed our transactions and any payments to each of these entities and found that these transactions and payments were made below the level set forth in applicable independence standards.
In addition to the above standards for director independence, each director who serves on the Audit Committee, Compensation and Human Capital Committee and Corporate Governance Committee satisfies the additional standards established by the SEC and NYSE, as applicable, for such committee membership.
Director Nomination Process
The Corporate Governance Committee makes recommendations to our Board for director nominations; identifies and screens potential new candidates, including by reviewing recommendations from other directors, management and shareholders; and assesses the ongoing contributions of incumbent directors whose terms are expiring, in each case with input from all other directors. The Corporate Governance Committee may also retain search firms to assist in identifying and screening candidates.
The Corporate Governance Committee will consider director candidates recommended by a shareholder in the same manner as all other candidates recommended by other sources. A shareholder may recommend a candidate at any time of the year by writing to our Corporate Secretary at the contact details set forth in “Other Information—Communicating With Us” below.
A shareholder, or group of up to 20 shareholders, owning 3% or more of our outstanding common stock continuously for at least three years, could submit director nominees for up to two individuals or 20% of our Board (whichever is greater) for inclusion in our Proxy Statement if the shareholder(s) and the nominee(s) meet the requirements in our Bylaws.
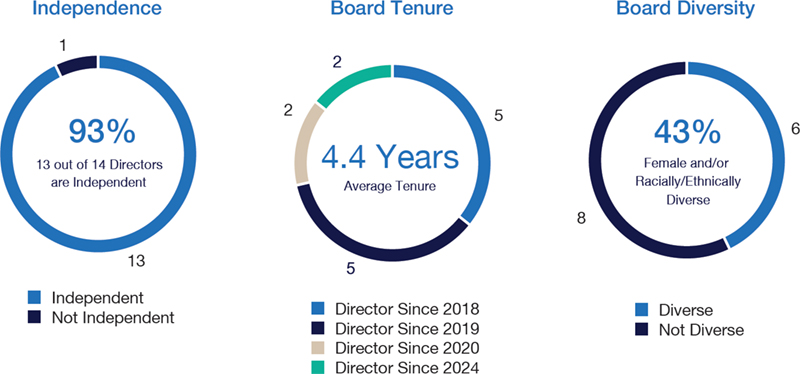
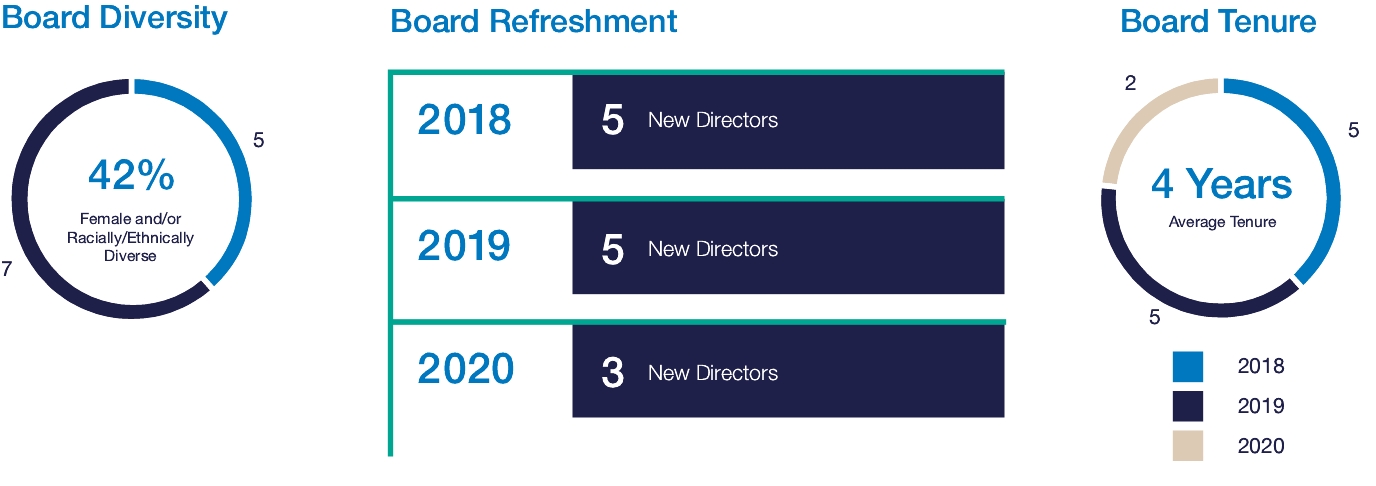
Board Diversity and Tenure
Our Board is committed to building a Board consisting of directors with diverse experiences and backgrounds. Our Corporate Governance Guidelines state that our Board will select director candidates who represent a mix of backgrounds and experiences that will enhance the quality of our Board’s deliberations and decisions and that Board membership should reflect diversity in its broadest sense, including persons diverse in geography, gender, race and ethnicity. Additionally, the charter of our Corporate Governance Committee states that the committee will actively consider for selection as directors those persons who are diverse in experience, ideas, gender, race and ethnicity.
Our Board is relatively short-tenured. Five of our directors joined our Board just prior to our initial public offering in September 2018, and five additional directors joined our Board in March 2019, when Eli Lilly exited its remaining ownership in Elanco. In December 2020, we enhanced our Board by adding additional directors to strengthen the financial and industry-specific expertise on our Board and to help drive innovation and improve our operations, and in 2024, we added two additional animal health industry executives to our Board. The four Class III directors we have nominated for re-election at the Annual Meeting have an average tenure of just under five years and our Board has an average tenure of approximately 4.4 years.
| 38 | 2024 Proxy Statement |  |
Our Corporate Governance Guidelines state that there is no limit on the number of terms for which a director may be elected and that our Board does not endorse arbitrary term limits on directors’ service. However, our Corporate Governance Guidelines also state that our Board does not believe in automatic re-nomination of directors and that the annual self-evaluation process described in “—Board Evaluations” below will be an important determinant for continuing service. Board Evaluations In the spirit of our values of excellence and continuous improvement, our Board is committed to regular Board and Committee assessments. This helps ensure that our Board’s governance and oversight responsibilities are well executed and updated to reflect best practices. At the end of each quarterly Board meeting, our Board meets in executive session, both with and without our Chief Executive Officer, to discuss whether the meeting’s objectives were satisfied and to identify issues that might require additional dialogue. Each of our Board’s standing committees also regularly meets in executive session for the same purposes. Our Board also conducts an annual self-evaluation process, which is led by the Chairman of our Board and the Corporate Governance Committee. Directors complete a comprehensive questionnaire evaluating the performance of our Board as a whole and the committees on which the director serves. The directors’ responses are aggregated and anonymized to encourage the directors to respond candidly and maintain the confidentiality of responses. The results are reviewed by the Corporate Governance Committee and summarized for the Board, which reviews the results in executive session. Each Board committee also separately reviews the feedback received for such committee in executive session. The Chairman then conducts one-on-one meetings with each director to discuss the evaluations and any other matters raised by the directors. We believe this annual evaluation process provides our Board and its committees with valuable insights regarding areas where our Board functions effectively as well as areas where our Board may improve. Recommendations for improvement derived from the annual evaluation process are used to adjust our Board’s future agendas and practices and responses to the evaluations also inform the topics for director education over the course of the following year. |
Related Party Transactions
Our Board has adopted a written policy, which is referred to as the “related person transaction policy,” for the review of any transaction, arrangement or relationship in which we are a participant and one of our executive officers, directors, director nominees or beneficial holders of more than 5% of Elanco’s total equity (or their immediate family members), each of whom is referred to as a “related Secretary person,” has a direct or indirect material interest.
If a related person proposes to enter into such a transaction, arrangement or relationship, which is referred to as a “related person transaction,” the related person must report the proposed related person transaction to our EVP, General Counsel and Corporate Secretary, who will report it to the Audit Committee. The policy calls for the proposed related person transaction to be reviewed and, if deemed appropriate, approved by the Audit Committee. In approving or rejecting such proposed transactions, the Audit Committee considers all relevant facts and circumstances. The Audit Committee will approve only those transactions that, in light of known circumstances, are deemed to be in ourthe Company’s best interests. In the event that any member of the Audit Committee is not a disinterested person with respect to the related person transaction under review, that member will be excluded from the consideration of such related person transaction; provided, however, that such Audit Committee member may be counted in determining the presence of a quorum at the meeting of the Audit Committee at which such transaction is acted upon.
| Elanco Animal Health Incorporated | 39 | 2024 Proxy Statement |
Business Ethics, Compliance and Privacy
We govern our business with the highest ethical standards, while creatingstandards; the way we operate rests on a space where all employees feel safe, respected, empowered and invested to make a difference in society. All Board members and employees have a shared responsibility to conduct businessfoundation of doing the right thing, the right way, every day. Our employees and directors have a shared responsibility for developing and cultivating a culture of integrity, respect and excellence. We encourage employees and third parties to report any known or suspected violations of our Code of Conduct, policies, procedures or laws that regulate our business without fear of retaliation. All concerns raised are timely and thoroughly investigated.
Elanco's Ethics & Compliance function led by our Chief Compliance Officer, is responsible for overall strategy, oversightour compliance program which includes global compliance policies and governance across five core areas: enterpriseprocedures, risk management, global ethicsassessments, anti-corruption due diligence, training and compliance, global anti-corruptioncommunications, and third-party management, global privacy and ethical interactions with external parties.conflicts of interest. Additionally, we have a global network of ethics and compliance partners that support program implementation and execution locally. Our Integrated Controls Committee, comprised of cross-functional senior leaders, provides program oversight of our ethics and compliance program. The Chief Compliance Officer provides quarterly reports to senior leadership and the Audit Committee and reports regularly to the Board.
Elanco is committed to the ethical management and processing of personal data related to our customers, consumers, employees and other individuals. We are transparent about how we process personal data and are intentional about protecting it, while being respectful of individuals’ privacy rights. We have standards, procedures and policies governing the collection, use, disclosure, transfer, storage, and retention of personal data. Elanco’s dedicated Global Privacy Office manages the privacy inquiries of our customers and employees executes Privacy Impact Assessments and ensures compliance with privacy laws and regulations globally. Elanco’s Global Privacy Notice, located on our public websites, explains how we collect, use, disclose, transfer and retain data, and provides individuals with information about how to exercise their privacy rights with Elanco.
Code of Conduct
The 10 ethical responsibilities set out in the Elanco Code of Conduct guide our decisions and relationships, establish our behavioral expectations and set the high standards against which we measure our performance. The Code of Conduct applies to our Chief Executive Officer, Chief Financial Officer and other senior financial anddirectors, executive officers as well as our Board and all other employees. It is available at www.elanco.com/en-us/about-us/governance/e-and-c. e-and-c by clicking on the "read more" button. In addition, we hold our suppliers and other third parties with whom we do business to similar standards which are contained in our recently revised Business Partner Code of Conduct.
We have also adopted a Financial Code of Ethics that contains the ethical principles by which our Chief Executive Officer, Chief Financial Officer and other financial officers are expected to conduct themselves when carrying out their duties and responsibilities. It is available atwww.elanco.com/en-us/about-us/governance/corporate, by clicking on the “Financial Code of Ethics” link. Any amendments to or waivers from the Elanco Code of Conduct or our Financial Code of Ethics will be disclosed on our website within the time period required by applicable law following the date of such amendment or waiver.
Other Information
We have adopted Corporate Governance Guidelines in accordance with the corporate governance rules of the NYSE, that serve as a flexible framework within which our Board and its committees operate. These guidelines cover a number of areas, including the role of our Board, Board composition, director independence, director selection, qualification and election, director compensation, executive sessions, key Board responsibilities, Chief Executive Officer evaluation, succession planning, risk management, Board leadership and
 |
Each of the above documents, along with the Elanco Code of Conduct and our Financial Code of Ethics, is available in print upon request to our Corporate Secretary through the means described in “Other Information—Communicating withWith Us” below.
| 40 | 2024 Proxy Statement |  |
Non-Employee Director Compensation | ||
Non-employee directors receive compensation for their service to our Board. As an Elanco employee, Mr. Simmons, our President and CEO, does not receive compensation for his service as a director.
The Compensation and Human Capital Committee annually reviews the total compensation of our Non-Employee Directorsnon-employee directors and each element of our Non-Employee Directornon-employee director compensation program. As part of this process, the Compensation and Human Capital Committee evaluates market data provided by its independent compensation consultant, Willis Towers Watson (“WTW”) and makes a recommendation to our Board. Our Board determines the form and amount of Non-Employee Directornon-employee director compensation after reviewing the Compensation and Human Capital Committee’s recommendation. Our Amended and Restated 2018 Elanco Stock Plan (the “2018 Stock Plan”) provides for an annual limit of $800,000 for all compensation paid to a Non-Employee Director.
SUMMARY OF 2023 NON-EMPLOYEE DIRECTOR 2022 ANNUAL COMPENSATION PROGRAM
| | Role | | | Cash ($) | | | Deferred Stock Units ($) | |
| | All Non-Employee Directors | | | 90,000 | | | 240,000 | |
| | Chairman of the Board | | | 150,000 | | | — | |
| | Chair of the Audit Committee | | | 25,000 | | | — | |
| | Chair of the Compensation and Human Capital Committee | | | 20,000 | | | — | |
| | Other Committee Chairs | | | 16,000 | | | — | |
Cash ($) | Deferred Stock Units ($) | ||
| All Non-Employee Directors | 90,000 | 240,000 | |
| Chairman of the Board | 150,000 | — | |
| Chair of the Audit Committee | 25,000 | — | |
| Chair of the Compensation and Human Capital Committee | 20,000 | — | |
| Other Committee Chairs | 16,000 | — |
Cash Retainers. Retainers. Our Non-Employee Directorsnon-employee directors each received an annual cash retainer of $90,000, the Chairman of our Board received an additional annual cash retainer of $150,000, the Chair of the Audit Committee received an additional annual cash retainer of $25,000, the Chair of the Compensation and Human Capital Committee received an additional annual cash retainer of $20,000 and the Chair of each of the three other standing committees of our Board received an additional annual cash retainer of $16,000. Through 2022, all cash retainers were paid in monthly installments. Starting in 2023, allAll cash retainers are paid in quarterly installments.
Equity-Based Awards.A substantial portion of each Non-Employee Director’snon-employee director’s annual retainer is in the form of equity awards. Pursuant to the Elanco Animal Health, Inc. Directors’ Deferral Plan (the “Directors’ Deferral Plan”), Non-Employee Directorsnon-employee directors are granted deferred stock units (“DSUs”) on or about November 30 of each year. Each DSU is the economic equivalent of one share of our common stock. These DSU awards are fully vested on the grant date and are subject to mandatory deferral under the terms of the Directors’ Deferral Plan until the second January following the recipient’s departure from service on our Board. In November 2022,2023, the number of DSUs underlying each DSU award was determined by dividing $240,000 (increased from $210,000) by the per share closing price of our common stock on the date of grant.
| Elanco Animal Health Incorporated | 41 | 2024 Proxy Statement |
Other Compensation.Our Non-Employee Directorsnon-employee directors may be reimbursed for reasonable out-of-pocket travel expenses incurred in connection with attendance at Board and committee meetings and other Board-related activities.
Directors’ Deferral Plan.Under the Directors’ Deferral Plan, a Non-Employee Director’snon-employee director’s annual award of DSUs is credited to a deferred stock account (as described below). The Directors’ Deferral Plan also allows Non-Employee Directorsnon-employee directors to defer receipt of all or part of their cash compensation until after their service on our Board has ended. Each director may choose to invest their deferred cash compensation in one or both of the following two accounts:
| ● | Deferred Stock Account.This account allows the director to invest the director’s cash compensation into a deferred common stock equivalent. Funds in this account are credited as hypothetical shares of our common stock based on the closing stock price on pre-set dates. The number of shares credited in respect of deferred cash compensation is calculated by the amount deferred divided by the closing stock price on pre-set dates. In addition, the annual equity award described above is credited to this account. The Company does not pay dividends but, if it did, deferred stock accounts would be credited with dividend equivalents. |
 |
Deferred Compensation Account. Funds in this account earn interest each year at a rate of |
Amounts under both accounts are paid in a lump sum in January of the second plan year following the plan year in which the director separates from service or in annual installments over a period between two to ten years (as selected by the director) beginning at the same time the lump sum payment would be made. Amounts credited to the director’s deferred stock account would generally be paid in shares of Elanco common stock and amounts credited to the director’s deferred compensation account would be paid in cash.
Stock Ownership Guidelines. Guidelines. Our Corporate Governance Guidelines state that directors should hold meaningful equity ownership positions in Elanco and that our Board will consider from time-to-time equity ownership requirements for Non-Employee Directors.non-employee directors. To help facilitate meaningful stock ownership, as described above, a significant portion of Non-Employee Directornon-employee director compensation is made in the form of company equity or equity equivalents, and as also noted above, the directors are unable to sell or otherwise transfer any of the shares granted to the directors by the Company until January of the second plan year after the director ceases to serve on the Board. Mr. Simmons, our President and CEO, is subject to and is compliant with the stock ownership guidelines applicable to our executive officers, as more fully described in “Compensation Discussion and Analysis—Governance and Other Matters—Stock Ownership and Holding Guidelines” below.
| Elanco Animal Health Incorporated | 42 | 2024 Proxy Statement |
2023 NON-EMPLOYEE DIRECTOR COMPENSATION
The following table shows information regarding the compensation earned or paid during 20222023 to Non-Employee Directorsnon-employee directors who served on our Board during the year. Mr. Simmons’ compensation is shown in the table entitled “Summary Compensation Table” and the related tables under “Executive Compensation Tables” below.
| | Name | | | Fees Earned or Paid in Cash ($) | | | Stock Awards ($)(1) | | | Total ($) | |
| | Kapila Anand | | | 115,000 | | | 240,013 | | | 355,013 | |
| | John Bilbrey | | | 106,000 | | | 240,013 | | | 346,013 | |
| | William Doyle | | | 90,000 | | | 240,013 | | | 330,013 | |
| | Scott Ferguson(2) | | | 67,500 | | | — | | | 67,500 | |
| | Art Garcia | | | 90,000 | | | 240,013 | | | 330,013 | |
| | Michael Harrington | | | 90,000 | | | 240,013 | | | 330,013 | |
| | Paul Herendeen | | | 90,000 | | | 240,013 | | | 330,013 | |
| | David Hoover | | | 256,000 | �� | | 240,013 | | | 496,013 | |
| | Deborah Kochevar | | | 106,000 | | | 240,013 | | | 346,013 | |
| | Lawrence Kurzius | | | 110,000 | | | 240,013 | | | 350,013 | |
| | Kirk McDonald | | | 90,000 | | | 240,013 | | | 330,013 | |
| | Denise Scots-Knight | | | 90,000 | | | 240,013 | | | 330,013 | |
| Name | Fees Earned or Paid in Cash ($) | Stock Awards ($)(1) | Total ($) | |
| Kapila Anand | 115,000 | 240,006 | 355,006 | |
| John Bilbrey | 106,000 | 240,006 | 346,006 | |
| William Doyle | 90,000 | 240,006 | 330,006 | |
| Art Garcia | 90,000 | 240,006 | 330,006 | |
| Michael Harrington | 90,000 | 240,006 | 330,006 | |
| Paul Herendeen | 90,000 | 240,006 | 330,006 | |
| David Hoover | 256,000 | 240,006 | 496,006 | |
| Deborah Kochevar | 106,000 | 240,006 | 346,006 | |
| Lawrence Kurzius | 110,000 | 240,006 | 350,006 | |
| Kirk McDonald | 90,000 | 240,006 | 330,006 | |
| Denise Scots-Knight | 90,000 | 240,006 | 330,006 |
| (1) | As discussed in “—Equity-Based Awards” above, on November 30, |
The following Elanco DSUs were outstanding as of December 31, 2022:2023: Ms. Anand, 44,374;69,950; Mr. Bilbrey, 54,344;74,718; Mr. Doyle, 25,956; Mr. Ferguson, 7,307;46,330; Mr. Garcia, 38,896;59,270; Mr. Harrington, 41,120;61,494; Mr. Herendeen, 25,956;46,330; Mr. Hoover, 69,736;101,690; Dr. Kochevar, 39,857;60,231; Mr. Kurzius, 58,131;88,456; Mr. McDonald, 39,857;60,231; and Ms. Scots-Knight, 40,844.61,218. These numbers include, where applicable, the annual equity grant discussed in “—Equity-Based Awards” as well as DSUs earned by directors who have elected to defer their cash compensation into Elanco shares.
 |
Executive Officers | ||
This section describes the experience and other attributes of our executive officers other than Mr. Simmons, whose biography can be found in “Proposal No. 1: Election of Directors—Our DiectorDirector Nominees and Other Continuing Directors” above.
Timothy Bettington
Mr. Bettington, 50, serves as our Executive Vice President, Corporate Strategy and Market Development. With more than 25 years in the animal health industry, Mr. Bettington joined Elanco in this role in 2023, after serving as EVP and President of U.S. Operations and Global Customer Experience at Zoetis, an animal health company, from January 2020 to November 2022. At Zoetis, he led U.S. Commercial operations, supporting the livestock and companion animal portfolios. Prior to that, Mr. Bettington served in roles with increasing responsibility over the course of his career, including as Global Head Commercial Excellence from 2015 to 2016 and North American Region Head of Commercial Operations from 2017 to 2019, both at Boehringer Ingelheim Animal Health, an animal health company, working both in the livestock and companion animal sectors. Mr. Bettington has worked in the U.S., Europe and Australia and has developed a deep expertise in sales excellence, marketing, strategy, and business integration. Mr. Bettington holds a bachelors degree in Agriculture from Western Sydney University and a master’s degree in Marketing Management from the Macquarie Graduate School of Management in Sydney, Australia.
Ramiro Cabral
Dr. Cabral, 52, serves as our Executive Vice President and President of Elanco International. He leads our international commercial operations, having assumed additional responsibility for our European operations in January 2022. Dr. Cabral joined Eli Lilly (our former parent company) in 1998 and has served in positions of increasing responsibility over the course of his career. These positions include Vice President and Head of Operations, Elanco EMEA from 2013 to July 2017, Vice President and Chief Marketing Officer of Elanco from August 2017 to June 2018 and Executive Vice President Elanco International and Global Customer Value from July 2018 to December 2018. Dr. Cabral earned his veterinary degree at UNICEN, Argentina in 1995 and his Master of Business Administration from Purdue University in 2005.
 |
Ellen de Brabander, Ph.D.
Dr. de Brabander, 61, serves as our Executive Vice President, Innovation and Regulatory Affairs. Dr. de Brabander joined Elanco in October 2021 and is responsible for our global research and development and regulatory affairs organization and as such is responsible for progressing and refilling the innovation pipeline. She is a highly experienced research and development executive with a proven background of accomplishments in animal health, life sciences and fast-moving consumer goods sectors. From March 2014 to October 2021, Dr. de Brabander served as senior vice president for research and development with PepsiCo, a global food and beverage company, most recently having company-wide responsibility for food safety, quality, regulatory and research and development digital transformation. Prior to PepsiCo, she led research and development organizations for Merial (now Boehringer Ingelheim Animal Health), Intervet (now Merck Animal Health) and DSM. Dr. de Brabander earned her doctorate (cum laude) in bio-organic chemistry from Leiden University in the Netherlands and completed her post-doctoral work in molecular biology at the Massachusetts Institute of Technology. She has lived in six different countries, is the co-author of more than 60 publications in scientific journals, holds 18 patents and has received multiple awards for her research, including the Golden Medal from the Royal Dutch Chemical Society in 2000 for Best Chemist Under 40.
David Kinard
Mr. Kinard, 57, serves as our Executive Vice President, Human Resources, Corporate Communications and Administration. Mr. Kinard brings valuable leadership experience within the pharmaceutical industry and years of managing human resources programs to this position. He assumed his current role in August 2020 and has led our Human Resources function since our spin-off from Lilly in 2018. Mr. Kinard joined Lilly in 1997 and served in a wide variety of positions, including as Vice President of Human Resources of Lilly International and as a human resources leader in other Lilly businesses and functions. He holds a bachelor’s degree in social science and broadcast communications and a master’s degree in organizational behavior from Brigham Young University.
James Meer
Mr. Meer, 54, serves as our Senior Vice President, Chief Accounting Officer. Prior to joining Elanco in September 2018, Mr. Meer served as the Chief Financial Officer of Healthx, Inc., a healthcare technology company, which he joined in June 2017. He served as Senior Vice President of Finance at Appirio, an information technology consulting company, from 2014 to 2017 and as Vice President and Corporate Controller at Salesforce (previously ExactTarget), a cloud-based software company, from 2011 to 2014. Prior to 2011, Mr. Meer held various financial, accounting and strategy positions at 3M (previously Aearo Technologies), Hillrom, Hillenbrand Industries and Ernst & Young LLP. Mr. Meer received his bachelor’s degree in accounting and business from Marian University and earned his Master of Business Administration from Xavier University.
Grace McArdle
Ms. McArdle, 45, serves as our Executive Vice President, Manufacturing and Quality, a position she has held since July 2023. Prior to this position, Ms. McArdle held roles of increasing responsibility at Elanco, including among others, Vice President Supply Chain Operations from March 2020 to July 2023, Executive Director of Manufacturing Strategy and Operational Excellence from May 2019 to March 2020, Senior Director Operational Excellence from April 2018 to May 2019, and Senior Director API External Manufacturing from July 2017 to April 2018. Ms. McArdle held roles at both Eli Lilly and Eli Lilly Ireland, which she first joined in September 2000. Ms. McArdle holds two master’s degrees in engineering – Biopharmaceutical Engineering from University College Dublin and Chemical Engineering from Queen’s University Belfast.
 |
Rajeev (Bobby) Modi
Mr. Modi, 43, serves as our Executive Vice President, U.S. Pet Health and Global Digital Transformation. Mr. Modi joined Elanco in this role in March of 2022, after serving as Vice President and General Manager of Enhancers and Components at Conagra Brands from May 2021 to April 2022. At Congara Brands he was responsible for the company’s substantial Grocery business, positioning it for profitable growth. Prior to that, he held numerous Vice President roles at J.M. Smucker Co. from 2012 to 2021. Mr. Modi holds a bachelor's degree in Chemical Engineering from the University of Cincinnati and a master’s degree in Marketing, Management Strategy and Finance from Northwestern University’s Kellogg School of Management.
Shiv O'Neill
Ms. O’Neill, 50, serves as our Executive Vice President, General Counsel, and Corporate Secretary, a position she has held since March 2024. Ms. O’Neill is responsible for the global strategy and operations of our legal team, including commercial, intellectual property, regulatory legal, labor & employment, M&A and business development legal, and the office of the corporate secretary. She also oversees Elanco’s ethics and compliance and ESG matters. Before her current role, Ms. O’Neill served as Interim General Counsel and Corporate Secretary from November 2023 to March 2024, as Vice President, Deputy General Counsel, Americas from May 2022 to November 2023, and as Vice President, Deputy General Counsel-US from October 2021 to May 2022. Prior to joining Elanco, Ms. O’Neill served as Senior Counsel at Cummins Inc., a multinational corporation that designs, manufactures, and distributes engines, filtration and power generation products, from February 2016 to October 2021. Prior to joining Cummins, Ms. O’Neill was a Partner at Baker & Daniels LLP (now Faegre Drinker Biddle & Reath LLP) from January 2010 to February 2016, and an Associate from March 2002 to December 2009. Ms. O’Neill started her career as in-house counsel at The Procter & Gamble Company. Ms. O’Neill received a Bachelor of Science from Loyola University Chicago and a Juris Doctor degree from the Indiana University Maurer School of Law.
José Manuel Correia de Simas, Ph.D.
Dr. Simas, 56, serves as our Executive Vice President, U.S. Farm Animal Business. Dr. Simas rejoined Elanco in this role in April 2020, after serving as President of Trouw Nutrition USA, an animal nutrition company, from 2018 to March 2020. In this role, he led its strategic change agenda to improve business quality and manufacturing productivity while building key capabilities in marketing, innovation, manufacturing and commercial excellence. Dr. Simas originally joined Elanco (then-owned by Eli Lilly) in 2000 as product manager for our Rumensin product, and throughout his career has served in key roles in the U.S. and internationally, including senior director of Latin America and Global Aquaculture, senior director of our U.S. Beef business and area director for Central, Eastern Europe, North Africa and Middle East. He received a bachelor’s degree from the Federal University of Lavras in Brazil and holds a master’s degree and a doctorate in animal nutrition and physiology from the University of Arizona, as well as a post doctorate from the University of São Paulo, Brazil.
Todd Young, JD
Mr. Young, 52, serves as our Executive Vice President and Chief Financial Officer, a position he has held since he joined Elanco in November 2018. Mr. Young oversees our financial operations, including our treasury, investor relations, tax functions, and commercial finance teams as well as information technology. He brings significant financial leadership experience in pharmaceutical and healthcare as well as a focus on strategic and commercial decision-making to his position. Prior to joining Elanco, Mr. Young served as Executive Vice President and Chief Financial Officer at ACADIA Pharmaceuticals Inc., a biopharmaceutical company, from August 2016 to October 2018, where he oversaw their financial functions as well as information technology and facilities. Prior to that, he served in roles of increasing responsibility at Baxter International Inc. and its spin-off company, Baxalta, a biopharmaceutical leader in hematology, immunology and oncology, most recently as Baxalta’s Senior Vice President and Treasurer. Mr. Young received his bachelor’s degree in economics from Grinnell College and a Juris Doctor from the University of Michigan.
 |
Proposal No. Ratification of of Independent Auditor | ||
The Audit Committee has reappointed Ernst & Young LLP (“EY”) as our independent registered public accounting firm for 2023.2024. EY has served as our independent auditor since 2017, when we were still a wholly owned subsidiary of Lilly.
Based on this year’s assessment of EY’s performance, the Audit Committee believes that the continued retention of EY to serve as our independent registered public accounting firm is in our best interests as well as those of our shareholders. EY has gained institutional knowledge and expertise regarding our global operations, accounting policies and practices and internal control over financial reporting, in particular through our separation from Eli Lilly in 2018 and our becoming an independent company. The Audit Committee believes that our audit and other fees are competitive with those of our peer companies in part because of EY’s familiarity with us and our operations.
At the Annual Meeting, our shareholders are being asked to ratify the appointment of EY as our independent registered public accounting firm for 2023.2024. Although ratification of this appointment is not required, we value the opinion of our shareholders and, in the event of a negative vote on this proposal, the Audit Committee will reconsider its selection. Even if this appointment is ratified, the Audit Committee may, in its discretion, appoint a different independent registered public accounting firm at any time during the year if the Audit Committee determines that such a change would be in our best interests.
Representatives of EY are expected to attend the Annual Meeting and will be available to respond to questions. Those representatives will have the opportunity to make a statement if they wish to do so.
Recommendation of the Board
| The Board unanimously recommends a vote “FOR” the ratification of the appointment of Ernst & Young LLP as our principal independent auditor for 2024. | ||
| 47 | 2024 Proxy Statement |  |
Fees Paid to Independent Registered Public Accounting Firm
The following table shows the fees incurred for services rendered on a worldwide basis by EY for 20222023 and 2021.2022.
| | | | 2022 ($) | | | 2021 ($) | | |
| | Audit Fees(1) | | | 11,561,448 | | | 11,509,055 | |
| | Audit-Related Fees(2) | | | 146,334 | | | 490,546 | |
| | Tax Fees(3) | | | 83,902 | | | 23,490 | |
| | All Other Fees | | | — | | | — | |
| | Total Fees | | | 11,791,684 | | | 12,023,091 | |
2023 ($) | 2022 ($) | | |
| Audit Fees(1) | 12,153,830 | 11,561,448 | |
| Audit-Related Fees(2) | 11,667 | 146,334 | |
| Tax Fees(3) | 5,103 | 83,902 | |
| All Other Fees | — | — | |
| Total Fees | 12,170,600 | 11,791,684 |
| (1) | Fees primarily related to professional services rendered in connection with the audit of Elanco’s annual consolidated and subsidiary financial statements and internal control over financial reporting, reviews of quarterly financial statements and audit services provided in connection with statutory and regulatory filings and audit procedures related to our acquisitions of |
| (2) | Fees primarily related to professional services that are reasonably related to the performance of the audit or review of Elanco’s financial statements, including services related to |
| (3) | Fees primarily related to tax compliance services. |
SERVICES PERFORMED BY THE INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The Audit Committee pre-approves all services performed by Elanco’s independent registered public accounting firm, in part to assess whether the provision of such services might impair the auditor’s independence. The Audit Committee’s policy and procedures are as follows:
| ● | Audit Services. The Audit Committee approves the annual audit and attestation services engagement and, if necessary, any changes in terms, conditions and fees resulting from changes in audit scope, company structure, or other matters. |
| ● | Audit-Related Services. Audit-related services are assurance and related services that are reasonably related to the performance of the audit or reviews of the financial statements and that are traditionally performed by the independent registered public accounting firm. The Audit Committee believes that the provision of these services does not impair the independence of the firm. |
| ● | Tax Services. The Audit Committee believes that, in appropriate cases, the independent registered public accounting firm can provide tax compliance services, tax planning and tax advice without impairing its independence. |
| ● | Other Services. The Audit Committee may approve other services to be provided by the independent registered public accounting firm if (i) the services are permissible under SEC and Public Company Accounting Oversight Board (“PCAOB”) rules, (ii) the committee believes the provision of the services would not impair the independence of the auditor and (iii) management believes that the auditor is the best choice to provide the services. |
| ● | Approval Process. At the beginning of each audit year, management requests prior committee approval of the annual audit, statutory audits and quarterly reviews for the upcoming audit year as well as any other services known at that time. Management will also present at that time an estimate of all fees for the upcoming audit year. As specific engagements are identified thereafter, they are brought forward to the committee for approval. |
For each engagement, management provides the Audit Committee with information about the services and fees, sufficiently detailed to allow the committee to make an informed judgment about the nature and scope of the services and the potential for the services to impair the independence of the firm. After the end of the audit year, management provides the Audit Committee with a summary of the actual fees incurred for the completed audit year.
| 48 | 2024 Proxy Statement |  |
Audit Committee Report
To the extent that this Proxy Statement has been or will be specifically incorporated by reference into any other filing of Elanco under the Securities Act of 1933, as amended (the “Securities Act”), or the Exchange Act, this section entitled “Audit Committee Report” will not be deemed to be so incorporated, unless specifically provided otherwise in such filing.
Four independent directors comprise the Audit Committee. The Audit Committee operates under a written charter adopted by our Board. In addition, our Board has determined that each of our Audit Committee members satisfy the financial expertise requirements of the NYSE and that the Chair of the Audit Committee, Ms. Anand, has the requisite experience to be designated as an “audit committee financial expert” as that term is defined by the rules of the SEC.
The Audit Committee reviews Elanco’s financial reporting process on behalf of our Board. Management has the primary responsibility for the financial statements and the reporting process, including the systems of internal controls and disclosure controls. In this context, the Audit Committee has met and held discussions with management and the independent auditor. Management represented to the Audit Committee that Elanco’s consolidated financial statements were prepared in accordance with GAAP and the Audit Committee has reviewed and discussed the audited financial statements and related disclosures with management and the independent registered public accounting firm, including a review of the significant management judgments underlying the financial statements and disclosures.
The independent registered public accounting firm reports to the Audit Committee, which has sole authority to approve and replace the firm (subject to shareholder ratification).
The Audit Committee has discussed with Elanco’s independent registered public accounting firm the matters required to be discussed with the Audit Committee by generally accepted auditing standards, the PCAOB and the NYSE, including the quality, not just the acceptability, of the accounting principles, the reasonableness of significant judgments and the clarity of the disclosures in the financial statements. In addition, the Audit Committee has received the written disclosures and the letter from the independent registered public accounting firm required by applicable requirements of the PCAOB regarding communications with the Audit Committee concerning independence and has discussed with the firm the auditor’s independence from Elanco and its management. In concluding that the auditor is independent, the Audit Committee determined, among other things, that the non-audit services provided by the auditor were compatible with its independence and were pre-approved. Consistent with the requirements of the Sarbanes-Oxley Act of 2002, the Audit Committee has adopted policies to ensure the independence of Elanco’s independent registered public accounting firm, such as prior committee approval of non-audit services and required audit partner rotation.
The Audit Committee discussed with Elanco’s internal auditor, chief compliance officer and independent auditors the overall scope and plans for their respective audits. The Audit Committee periodically meets with the internal and independent auditors, with and without management present and in private sessions with members of senior management (such as the chief financial officer and the chief accounting officer) to discuss the results of their examinations, their evaluations of Elanco’s internal controls and the overall quality of Elanco’s financial reporting. The Audit Committee also meets at least quarterly in executive session.
Based on the reviews and discussions described in this report and subject to the limitations on the roles and responsibilities of the Audit Committee referred to above and in its charter, the Audit Committee recommended to our Board that the audited consolidated financial statements of Elanco be included in Elanco’s Annual Report on Form 10-K for the fiscal year ended December 31, 2022,2023, for filing with the SEC.
Respectfully submitted,
Kapila K. Anand (Chair)
John P. Bilbrey
Art A. Garcia
Paul Herendeen
| 49 | 2024 Proxy Statement |  |
Proposal No. Advisory Vote to Approve Executive Compensation | ||
As required by Section 14A of the Exchange Act, our shareholders are being asked to approve, on an advisory basis, the compensation of our named executive officers (“NEOs”), as disclosed pursuant to the SEC’s compensation disclosure rules in the sections entitled “Compensation Discussion and Analysis” and the section entitled “Executive Compensation Tables,” which include both the executive compensation tables and the accompanying narrative disclosure.
Our executive compensation programs are designed to help achieve the goals of attracting, engaging and retaining highly talented individuals who are committed to our core values of integrity, excellence and respect for people, while balancing the long-term interests of shareholders and customers. The Compensation and Human Capital Committee, which consists entirely of independent directors, has examined our executive compensation program and believes it aligns with our compensation philosophy and objectives as well as the pay practices of our peer group. The Compensation and Human Capital Committee has also determined that the specific pay decisions for the NEOs are appropriate given our performance, the executives’ contributions and our shareholders’ interests.
While this vote is advisory and non-binding, our Board and the Compensation and Human Capital Committee value the opinion of our shareholders and will take into account the voting results when making future compensation decisions. We currently ask our shareholders to vote on NEO compensation on an annual basis.
We encourage shareholders to read the sections entitled “Compensation Discussion and Analysis” and “Executive Compensation Tables” for more information about the details of our executive compensation program and the decisions made by the Compensation and Human Capital Committee in 2022.2023.
Recommendation of the Board
| The Board unanimously recommends a vote | ||
| 50 | 2024 Proxy Statement |  |
Compensation Discussion and Analysis | |||||
This sectionCompensation Discussion and Analysis (“CD&A”) describes the compensation program applicable to our NEOs and their compensation thereunder in 2022,2023, including a description of our compensation philosophy, the elements of each program, the factors that the Compensation and Human Capital Committee (referred to as the “Committee” for purposes of this CD&A) considered in setting NEO compensation and how our financial results affected payouts to each NEO for service in 2022. 2023.
The NEOs for 20222023 are:
Jeffrey Simmons | ||||||
Todd Young | ||||||
Ellen de Brabander | ||||||
Ramiro Cabral | ||||||
2023 Performance and the economic slowdown impacting Europe and U.S. pet retail, as well as competitive innovation.Results
Elanco ended 2023 with momentum, returning to constant currency revenue growth for the full year. In 2023, we laid the groundwork for actions to improve our performance, leading to decisive actions in early 2024, which allow us to strengthen our focus and concentrate investment on the most significant value creation opportunities in pet health and livestock sustainability. Performance and results in 2023 and early 2024 include: ● Demonstrated momentum of our Innovation, Portfolio and Productivity strategy to deliver long-term shareholder value ● Announced an agreement in 2024 to divest our aquaculture business for $1.3 billion, with plans to use after-tax proceeds for debt paydown ● Announced strategic restructuring in 2024 to reinvest in more significant value creation opportunities ● Revenue was | |||||
1% ◦ Revenue from new products launched since 2021 was $275 million, more than double the contribution in 2022 ● Our GAAP net loss was | |||||
increased long-term treasury rates in the third quarter of 2023 ● Gross margin | |||||
planned reduced throughput of certain manufacturing sites in support of efforts to reduce inventory balances on-hand and improve cash conversion 22.2%● Adjusted EBITDA2 was ● Adjusted EPS2 was $0.89, a year-over-year decline of $0.22 driven primarily by the impact of higher interest expense, higher taxes and the unfavorable impact of foreign exchange rates | |||||
(1) Constant currency is a non-GAAP measure, representing revenue growth excluding the impact of foreign exchange rates.
(2) Non-GAAP financial measures. See Appendix A to this Proxy Statement for a description and reconciliation of GAAP to non-GAAP measures.
 |
Executive Compensation Philosophy and Practices
The purpose of our compensation philosophy is to ensure alignment between the Company, our executives and shareholders. Our compensation program is designed to achieve the following three objectives:
| ● | Attract, retain, motivate and reward top growth. |
| ● | Pay for paid. |
| ● | Create sustained, long-term shareholder shareholders. |
We achieve these objectives by:
| ● | Providing an executive compensation program that includes base salaries, annual cash incentives and equity incentives similar to programs provided to Elanco’s other employees or similarly situated executives in a competitive talent market. |
| ● | Targeting compensation, in aggregate, at the median (50th percentile) of the competitive market, which is comprised of similarly sized companies within the life sciences industry, with consideration of other industries, as appropriate. |
| ◦ | In certain situations, where there is scarcity of talent for a critical role and there is a need to offer a more competitive compensation package to attract such scarce talent, we may exceed the targeted median positioning of the market. |
| Designing a compensation program that is more heavily weighted toward equity, as compared to the median of the market for allocation between equity and cash, to promote an ownership mentality and ensure shareholder alignment. |
| ● | Promoting a team-based approach through the alignment of pay with Elanco’s results while enabling leadership to differentiate pay throughout the year to recognize performance. |
| ● | Requiring that senior executives maintain a meaningful stock ownership interest to align their financial interests with those of shareholders. |
| ● | Limiting perquisites and other non-performance-based elements of compensation. |
| ● | Ensuring the program does not incentivize excessive risk taking. |
| ● | Considering shareholder feedback through annual say-on-pay results, regular shareholder engagement and other sources when designing our compensation and benefit program. |
| ● | Designing the program with consideration of the industry in which we operate and the impact of market conditions. |
| Elanco Animal Health Incorporated | 52 | 2024 Proxy Statement |  |
In addition to strong alignment of pay with our performance, we maintain sound governance practices, including the following:

Executive Compensation Process
We seek to maintain a market-competitive executive compensation program that is consistent with our compensation philosophy and attracts and retains executives who perform at a high level and contribute to our success.
The Compensation and Human Capital Committee, which consists entirely of independent directors, determines our compensation philosophy and executive compensation program design and is the decision-making body on all matters relating to the compensation paid to the NEOs. The Compensation and Human Capital Committee recognizes that at the time of our initial separation from Lilly, most of our executive officers received compensation at levels substantially below our peer group median compensation level relative to their roles in a newly independent public company. This discrepancy became more acute after our acquisition of Bayer Animal Health when the size and complexity of the Company dramatically increased. In order to smooth the impact on our executive compensation budget and at the advice of the Compensation and Human Capital Committee’s independent compensation consultant, the committee utilized a “glidepath” approach to gradually bring these executives closer to market median compensation over a several-year period. The Compensation and Human Capital Committee continues to adjust the compensation of our NEOs along this glidepath because our NEOs continue to lag behind the median compensation level of our peer group.
For 2022,2023, each NEO’s compensation was set, in part, based on an assessment of the NEO’s individual performance against achievement of objectives established at the start of the year. “Individual performance” included the NEO’s demonstration of our values and leadership behaviors. Company performance was considered in two ways: (1) in setting 20222023 compensation, a comparison to overall performance in 20212022 based on a variety of metrics and (2) in determining achievement of 20222023 bonus compensation, measured against specific goals established at the beginning of the year. We consider peer group data in a benchmarking review. After this benchmarking review, the Compensation and Human Capital Committee attempts to establish compensation relative to the peer group median for each NEO. However, consistent with our compensation philosophy, the Compensation and Human Capital Committee can differentiate pay levels from those at peer companies based on the results of the aforementioned assessments as well as other factors the Compensation and Human Capital Committee believes are relevant, including, but not limited to, scarcity of talent for a critical role. See “—Compensation Benchmarking” below for more information about how the Compensation and Human Capital Committee uses peer group data in making executive compensation decisions.
| Elanco Animal Health Incorporated | 53 | 2024 Proxy Statement |
At the time of our initial separation from Eli Lilly and our subsequent acquisition of Bayer’s animal health business, most of our executive officers received compensation significantly below our peer group medians due to their roles in a newly independent public company. At this time, the Committee utilized a “glidepath” approach to gradually bring these executives to market. In 2023, we reached the culmination of this glidepath trajectory for Elanco’s tenured executive officers, with all officers receiving target total compensation at or near peer median levels. The Committee may use a similar glidepath approach for future executive officers who are new in role and are still establishing their individual performance record.
Finally, the Compensation and Human Capital Committee determines the specific metrics to be used to measure company performance for purposes of the annual cash incentive program and long-term performance share awards. The specific metrics
 |
ROLE OF SHAREHOLDERS
|
| ● | Selection of Performance Measures for Incentive Compensation – in 2022, we transitioned to Elanco Cash Earnings (also known as “ECE”), a metric similar to economic value added, for our incentive programs to further incentivize our productivity efforts and reinforce a culture of ownership. The Committee acknowledges shareholder sentiment that may prefer use of multiple metrics; however, the Committee believes that the culmination of many metrics into ECE operates to incentivize both growth and return on capital invested in our business that is appropriate for execution of our current strategy. |
| ● | Base Salary Increases – as described above, the Committee has utilized a “glidepath” approach to align our executives’ compensation to market to address compensation levels that were below median when set in connection with our separation from Lilly and our subsequent acquisition of Bayer’s animal health business. The changes made to base salary in recent years have now closed the structural compensation gap as of the changes made for 2023. Going forward, base salary changes will be considered consistent with our philosophy of targeting a range around median, taking other factors into consideration as described below. |
| Elanco Animal Health Incorporated | 54 | 2024 Proxy Statement |
| ● | Two-Year Performance Period for Performance Awards and Use of Single, Absolute Metric – the performance awards granted in 2023 continued to have a two-year performance period with an Adjusted EBITDAR performance measure. The Committee believes the two-year performance period remains appropriate in light of the rate of transformation since our separation from Lilly and is committed to re-evaluating this as a more consistent pattern of performance is established. The Committee continues to believe that Adjusted EBITDAR, with each year’s performance targets set relative to the prior year’s results, focuses on a key metric that is the culmination of many metrics and encourages efficient use of assets and a long-term ownership mindset. |
While we believe the specific target performance levels are competitively sensitive during the performance period, we remain committed to transparent disclosure of the performance measures at the end of each performance period. The Committee remains committed to setting rigorous performance goals. Payouts across our annual incentive awards and performance awards have ranged from below target to above target, suggesting that the performance goals reflect this rigor. Finally, performance awards remain the most recent say-on-pay vote,heavily weighted component of long-term incentive compensation, comprising 50% of the Compensation and Human Capitaltotal value of long-term incentive awards.
The Committee decidedcontinues to not make any changes tocarefully evaluate the design of our compensation program. program, including consideration of the factors above, and is guided by the Board’s strategy for long-term shareholder value creation and taking shareholder feedback into consideration.
For more information about our shareholder engagement efforts, see “Corporate Governance—Shareholder Outreach” above.
ROLE OF THE COMPENSATION CONSULTANT
The Compensation and Human Capital Committee directly engages an independent compensation consultant, WTW, that reports directly to the chair of the Compensation and Human Capital Committee. The consultant meets regularly and as needed with the Compensation and Human Capital Committee and has direct access to the chair during and between meetings. Among other duties, the consultant advises the Compensation and Human Capital Committee on competitive pay practices, assists with the determination of the peer group for compensation purposes and provides and analyzes compensation levels relative to market benchmarks. During 2022,2023, WTW performed no other services for us or our executive officers.
The Compensation and Human Capital Committee has the sole authority to retain and terminate its independent compensation consultant and approve its fees, which the Company pays. The Compensation and Human Capital Committee also considers the independence of the consultant in accordance with SEC and NYSE rules.
ROLE OF THE CHIEF EXECUTIVE OFFICER AND SENIOR MANAGEMENT
The Compensation and Human Capital Committee also works with Mr. Simmons, our Chief Executive Officer, as well as our Executive Vice President, Human Resources, Corporate Communications and Administration and other members of senior management, to oversee our executive compensation program. At the Compensation and Human Capital Committee’s request, Mr. Simmons plays a key role in the process as it relates to executive officers other than himself. Forhimself, including the NEOs other than Mr. Simmons, he:following:
| ● | recommends adjustments to annual base salaries and target annual cash incentive amounts; |
| ● | recommends equity incentive awards under our long-term incentive plan; and |
| ● | prepares an evaluation of each executive officer, including achievement of set objectives. |
The Compensation and Human Capital Committee considers Mr. Simmons’ evaluation and his direct knowledge of each NEO’s performance and contributions when making compensation decisions. However, Mr. Simmons is not present during Compensation and Human Capital Committee voting or deliberations regarding his own compensation and the Compensation and Human Capital Committee solely determines and approves (subject to ratification by the independent members of our Board) each element of Mr. Simmons’ compensation.
| 55 | 2024 Proxy Statement |  |
COMPENSATION BENCHMARKING
To provide the appropriate context for executive pay decisions, the Compensation and Human Capital Committee, in consultation with WTW, assesses the compensation practices and pay levels of our peer group. In general, the committeeCommittee uses the peer group to benchmark:
| ● | Base Salary |
| ● |
| ● | Long-Term Incentives (equity) |
| ● | Executive Program Design |
| ● | Industry Trend Analysis |
For 20222023 executive compensation benchmarking, the Compensation and Human Capital Committee used a peer group selected in 2021 whose2022 with median revenues were broadly similar to ours after our acquisition of Bayer Animal Health, with revenue of roughly$5.2 billion, generally ranging from one-half to two times Elanco’s revenue, with some companies having greater revenue but are in very comparable businesses.revenue. The Compensation and Human Capital Committee also believed these companies share important characteristics with us, such as being our direct competitors, operating in a similar business model, or employing people with the unique skills required to operate an established biopharmaceutical company like ours.
Based on the advice of WTW, the following group of 1917 companies was identified by the Compensation and Human Capital Committee as our peer group for 20222023 executive compensation benchmarking.
| Agilent Technologies, Inc. | Regeneron Pharmaceuticals, Inc. | |||||||
Baxter International Inc. | ||||||||
| Boston Scientific Corporation | IDEXX Laboratories, Inc. | |||||||
Vertex Pharmaceuticals, Inc. | ||||||||
| Charles River Laboratories International, Inc. | Zimmer Biomet Holdings, Inc. | |||||||
| DENTSPLY SIRONA Inc. | ||||||||
| Edwards Lifesciences Corporation | Perrigo Company plc | |||||||
The Compensation and Human Capital Committee periodically reviews our peer group and adds or removes companies in the peer group when appropriate to help ensure companies in the peer group are similar in size to us and appropriately reflect how we conduct our business. In August 2022,2023, the Compensation and Human Capital Committee revised the peer group to remove AlexionBaxter International, Boston Scientific Corporation, and Regeneron Pharmaceuticals because their revenues were above the high end of the screening criteria. The Committee added Catalent, Inc., QuidelOrtho Corporation, and Varian Medical Systems – both of which were acquired –Revvity, Inc. because they are in comparable industries (life sciences) and Endo International because it recently filed for bankruptcy. The committee added Horizon Therapeutics because it is in ahave comparable industry (biotechnology); has comparable revenue, was identified as being a peer of Elanco by a shareholder services organization and has an innovation focus.revenue. This updated peer group of 17 companies will be the peer group used for 20232024 executive compensation benchmarking.
| Elanco Animal Health Incorporated | 56 | 2024 Proxy Statement |
2023 NEO COMPENSATIONCompensation
Our executive pay program consists of three primary components:
ANNUAL BASE SALARY
| ● | Within a range of competitive pay and targeted to the median of the peer group. Base salaries are based on each person’s level of contribution, responsibility, and expertise, and are competitive with respect to base salaries in our peer group. |
| ● | To ensure that we can attract, retain, motivate and reward a leadership team that will |
ANNUAL CASH INCENTIVE
| ● | Performance based and determined by achievement of a change in Elanco Cash Earnings (as defined below) from the prior year. |
| ● | To provide focus and reward for the achievement of |
|  |
LONG-TERM EQUITY INCENTIVE AWARDS |
| ● |
| ● | Restricted Stock Units (“RSUs”) - 25% of total equity incentive award opportunity: These awards have the potential to vest in roughly one-third increments on each of the first three annual anniversaries of the grant date, subject to continued employment with us. |
| ● | Stock Options - 25% of total equity incentive award opportunity: These awards have the potential to vest in roughly one-third increments on each of the first three annual anniversaries of the grant date, subject to continued employment with us. |
| ● | The objective of these vehicles is to provide an incentive for delivering long-term shareholder value and to align the interests of executives and shareholders over the long-term. |
Specifically, the Compensation and Human Capital Committee is dedicated to ensuring that a substantial portion of executive compensation is “at-risk” and variable. A substantial majority of the NEOs’ total target direct compensation is directly affected by our company-wide performance. The charts below show the percentage of 20222023 fixed (base salary) and variable (annual cash bonus and equity award) for our CEO and an average of the other NEOs at target.
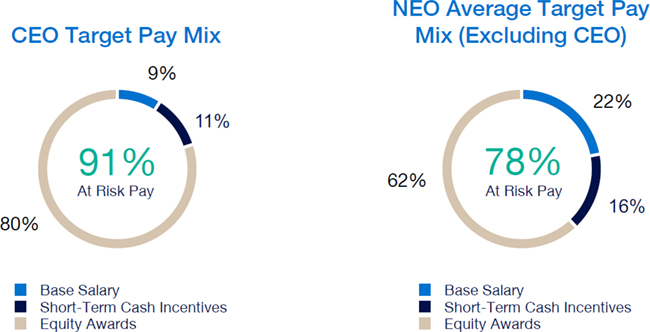
| Elanco Animal Health Incorporated | 57 | 2024 Proxy Statement |
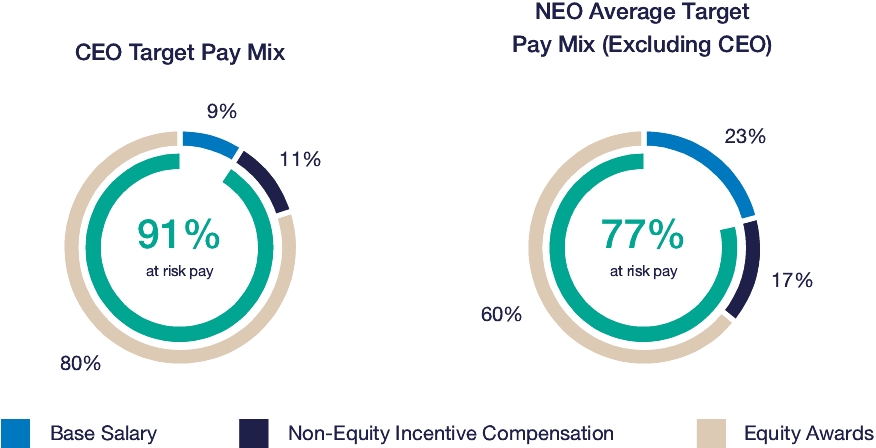
2023 ANNUAL BASE SALARY
During 2022,2023, base salaries for the NEOs were increased on average by 8%5%. These increases were established based on several factors, including peer group benchmarking, general external trends and experience in their roles. Base salaries were increased to align withFollowing the annual base salary increases for 2023, Elanco’s tenured executive officers now all receive compensation at or near peer median peer group benchmarks as a culmination of the several-year glidepath exercise to align executive compensation with market peers. This glidepath approach began after our separation from Lilly and was revised due to our updates to our peer group since the acquisition of Bayer Animal Health in 2020.levels.
| | Name | | | 2022 Annual Base Salary ($) | | | 2021 Annual Base Salary ($) | |
| | Mr. Simmons | | | 1,125,000 | | | 1,046,000 | |
| | Mr. Young | | | 639,000 | | | 580,000 | |
| | Dr. de Brabander(1) | | | 667,819 | | | 709,672 | |
| | Dr. Cabral | | | 572,000 | | | 475,000 | |
| | Mr. Modi(2) | | | 525,000 | | | — | |
| Name | 2022 Annual Base ($) | 2023 Annual Base ($) | Annual Change |
| Mr. Simmons | 1,125,000 | 1,200,000 | 6.7 % |
| Mr. Young | 639,000 | 661,400 | 3.5 % |
| Dr. de Brabander(1) | 667,819 | 709,748 | 6.3 % |
| Dr. Cabral | 572,000 | 592,000 | 3.5 % |
| Mr. Bettington(2) | — | 620,000 | — |
| (1) | Dr. de Brabander’s compensation is paid in Euros and was 656,200 and 634,000 Euros in 2023 and |
| (2) | Mr. |
|  |
2023 ANNUAL CASH INCENTIVE
In 2023, the Committee again selected Elanco Cash Incentive
ECE encourages and rewards the NEOs for making decisions that benefit the business and its shareholders over the prior year. While ECE is a short-term metric, the cumulative effect of positive ECE over the long-term creates shareholder value. If ECE improves, shareholder value has been created. The Compensation and Human Capital Committee believes that this metric better aligns with our growth and value creation strategy, which is to drive innovation over relatively long product cycles through ongoing prudent investments in research and development (“R&D.&D”). By requiring our business to earn more than our cost of capital on our investments on an annual basis over time, the Compensation and Human Capital Committee believes leadership will be further incentivized to invest in and deliver profitable innovation to drive revenue growth, prudently manage expenses, efficiently use our assets and otherwise take actions designed to create long-term, sustainable shareholder value. This is also designed to create a more significant ownership mentality within Elanco by further aligning management’s interests with those of our shareholders. To ensure our calculated capital charge of 8.5% creates rigorous accountability that is aligned with our core business model, we use a base of “Gross Investments.” We define Gross Investments as (i) an average of our gross operating assets over the four calendar quartersKey elements of the ECE calculation include:
| ● | Gross Investments - To ensure our calculated capital charge of 8.5% creates rigorous accountability that is aligned with our core business model, we use a base of “Gross Investments.” We define Gross Investments as (i) an average of our gross operating assets over the four calendar quarters of the performance year combined with (ii) the prior eight years, including the performance year, of adjusted R&D expenses. |
| ● | Gross Operating Assets - Using gross operating assets in our definition of Gross Investments, which excludes depreciation, ensures that year-over-year Adjusted EBITDAR growth is required to exceed our cost of capital. |
| ● | Adjusted EBITDAR – Adjusted EBITDAR starts with Adjusted EBITDA as we reflected in our financial reporting, and then adds back R&D expenses. A description and reconciliation of Adjusted EBITDA is included in Appendix A to this Proxy Statement. The add back of R&D expenses in detailed in the 2023 ECE calculation section below. |
| Elanco Animal Health Incorporated | 58 | 2024 Proxy Statement |
| ● | R&D Expenses - Categorizing our R&D expenses as investments required to drive long-term value, rather than as short-term costs, reinforces the commitment to consistently deliver pipeline milestones over time and strengthens the ownership mindset. We utilize eight years of prior R&D expense to closely align our Gross Investment base with our product cycle timing. |
As a result, because ECE requires Adjusted EBITDAR growth to achieve our target ECE and a payout at target, we consider target attainment to be rigorous. Payouts for achievement above or below target are based on linear interpolation, which rewards ECE growth above the prior eight years, includingyear, but pays out less than target for decreases in ECE versus the prior year. The establishment of the target and slope of the curve are both objective, quantitative metrics based on prior year performance year,and not based on an annual planning process, which aligns closely with shareholder interest and allows management to focus on achieving long-term sustainable growth.
The calculation of adjusted R&D expenses. Using gross operating assets, which excludes depreciation, ensures that year-over-year EBITDAR growthECE described above is required to exceed our cost of capital. Categorizing our R&D expenses as investments required to drive long-term value, rather than as short-term costs, reinforces the commitment to consistently deliver pipeline milestones over time and strengthens the ownership mindset. We utilize eight years of prior R&D expense to closely align our Gross Investment base with our product cycle timing.
| | We multiply the sum of our average Gross Operating Assets and eight years of Adjusted R&D expense (together, “Gross Investments”) by 8.5% to determine the Capital Charge on Gross Investments. | | | Subtracting the Capital Charge from GCE gives us ECE, which tells us whether we are generating a return greater or less than the Required Rate of Return of 8.5%. | | ||||
This calculation of ECE is presented in the following formula:
| | Adjusted EBITDA | | | Average Gross Operating Assets | |||
| + | | Adjusted R&D Expense | | + | | 8 years of Adjusted R&D Expense | |
| | | | Gross Investments | ||||
| - | | Taxes | | x | | Required Rate of Return | |
| | Gross Cash Earnings | - | | | Capital Charge on Gross Investments |
Each NEO was eligible to and did participate in our annual cash incentive program for 2022.2023. The program objective was linearly interpolated for achievement between threshold, target, and maximum goals of 0%, 100%, and 200%, respectively.
Our annual cash incentive program allows the Compensation and Human Capital Committee to make adjustments based on the effects of acquisitions, divestitures, restructurings or special charges or gains, changes in corporate capitalization, accounting changes,
 |
| Elanco Animal Health Incorporated | 59 | 2024 Proxy Statement |
Bonus targets for 20222023 and 20212022 are shown in the table below as a percentage of the NEO’s base salary.
| | Name | | | 2022 Bonus Target (%) | | | 2021 Bonus Target (%) | |
| | Mr. Simmons | | | 130 | | | 125 | |
| | Mr. Young | | | 80 | | | 75 | |
| | Dr. de Brabander | | | 70 | | | 70 | |
| | Dr. Cabral | | | 70 | | | 70 | |
| | Mr. Modi(1) | | | 70 | | | — | |
| Name | 2022 Bonus Target (%) | 2023 Bonus Target (%) | |
| Mr. Simmons | 130 | 130 | |
| Mr. Young | 80 | 80 | |
| Dr. de Brabander | 70 | 70 | |
| Dr. Cabral | 70 | 70 | |
| Mr. Bettington(1) | — | 70 |
| (1) | Mr. |
2023 BONUS PAYMENTS
In the second half of 2023, the Committee became aware of several non-operational factors impacting the Gross Investments calculation portion of ECE. In aggregate, all changes reduced our ECE by $5 million, lowering our corporate bonus payout by 1%. To better reflect the Committee’s original intent when it selected the ECE metric, the Committee elected to normalize the capitalized R&D spend portion of Gross Investments for the years prior to the Bayer Animal Health acquisition. Due to the eight-year time horizon of capitalized R&D expense included in Gross Investments, evaluating pre-Bayer R&D expense on a pro forma basis ensures that the business is appropriately incentivized for managing R&D spend. Without this adjustment, we would continue to show increases in R&D spend for the duration of the eight-year period after the Bayer acquisition due to global processing deadlinesthe relative size of the two entities, regardless of how effectively we managed our R&D spend. Because the cash incentive is based on the year-over-year change in ECE, and its custom,the normalization is related to pre-acquisition periods, this change was applied to both the prior and current year performance periods to appropriately normalize the R&D spend for ECE purposes. This change will continue to apply until eight years after the completion of the Bayer acquisition, which is 2028. A reconciliation of the 2022 ECE baseline after adjusting for this change is outlined in the table below.
2022 Adjusted ECE Baseline Normalized for Pre-Acquisition Bayer Animal Health (BAH) R&D Spend
(All numbers $M except %s)
| Average Gross Operating Assets | 15,672 | |||||||
| + 8 Years of Adjusted R&D Expense | 2,268 | |||||||
| Adjusted EBITDA | 1,023 | + Pre-Acquisition BAH R&D Adjustment | 910 | |||||
| + | Adjusted R&D Expense(1) | 310 | Gross Investments | 18,850 | ||||
| - | Taxes | (147) | x Required Rate of Return | 8.5 % | ||||
| Gross Cash Earnings | 1,186 | - Capital Charge on Gross Investments | (1,602) | = | ECE (416) Million |
| (1) | Adjusted R&D expense in the ECE calculation excludes depreciation and amortization to align with the Adjusted EBITDA calculation that excludes such items. |
In addition to the normalization of R&D spend related to the Bayer acquisition, the Committee made several other changes to the Gross Investments definition to eliminate non-operational factors influencing ECE. The Committee determined that ECE would be calculated exclusive of balance sheet valuation changes related to several items - foreign currency rates on goodwill and intangibles, impairments to goodwill, and interest rate-driven changes in accrued pension liabilities – all of which are non-cash items not associated with core business performance and generally not within management’s control. The Committee made this change to the definition of Gross Investments for both the 2023 performance period as well as all future years to provide consistency and avoid both undue windfalls or penalties to incentive pay.
| Elanco Animal Health Incorporated | 60 | 2024 Proxy Statement |
An itemized breakdown of the changes to the Gross Investments definition, and their resulting impact on ECE and the corporate bonus multiplier, are captured in the table below.
2023 Impact of Gross Investments Changes
| Adjustment Category | ECE Impact ($M) | Bonus Multiple Impact (%) |
| FX and Impairments to Goodwill | (35) | (10) |
| FX on Gross Intangibles | 16 | 5 |
| Rate Changes on Accrued Pension | 5 | 2 |
| Pre-BAH Normalized Capitalized R&D | 9 | 2 |
| Total Adjustments | (5) | (1) |
The Committee calculated ECE based upon the Company’s financials released on February 21, 2023 with our earnings release for the fourth quarter and full year of 2022. Subsequent to that release, an immaterial change was made to those financials which was included in our Annual Report on Form 10-K for the year ended December 31, 2022.2023. The 20222023 calculation as set forth below will be used as the base for the determination of the change in ECE when determining the bonus for 2023.2024.
20222023 ECE Calculation
(All numbers $M except %s)
| | | | | | | | | | | Average Gross Operating Assets | | | 15,672 | | | | | |||||||
| | | Adjusted EBITDA(1) | | | 1,023 | | | | | + | | | 8 Years of Adjusted R&D Expense | | | 2,268 | | | | | ||||
+ | | | Adjusted R&D Expense(2) | | | 310 | | | | | | | Gross Investments | | | 17,940 | | | | | ||||
- | | | Taxes | | | (147) | | | | | x | | | Required Rate of Return | | | 8.5% | | | | | |||
| | | Gross Cash Earnings | | | 1,186 | | | - | | | | | Capital Charge on Gross Investments | | | 1,525 | | | = | | | ECE ($339) million |
| | | Average Gross Operating Assets | 16,226 | ||||||
| Adjusted EBITDA | 979 | + | | 8 Years of Adjusted R&D Expense(2) | 3,130 | ||||
| + | Adjusted R&D Expense(1) | 317 | | Gross Investments | 19,356 | ||||
| - | Taxes | (143) | x | | Required Rate of Return | 8.5% | |||
| Gross Cash Earnings | 1,153 | - | | Capital Charge on Gross Investments | 1,645 | = | ECE (492) Million |
| (1) |
| Adjusted R&D expense in the ECE calculation excludes depreciation and amortization to align with the Adjusted EBITDA calculation that excludes such items. |
| (2) | Eight years of capitalized R&D expense includes normalization for pre-acquisition BAH expenses as explained above. |
The 20222023 ECE was ($339)492) million, a change of ($27)76) million versus the ECE of ($312)416) million in 2021. The primary contributing factor to the reduction in 2022 ECE was lower year-over-year Adjusted EBITDA.2022.
The payouts are linearly interpolated frombased on the minimum payout of 0 to thefollowing threshold, target and maximum of 200%.levels. Our 20222023 change in ECE of ($27) million resulted in a corporate bonus multiple of 93%79% of the target payout opportunity.opportunity, as reflected below:
| Threshold | Target | Maximum | 2023 Actual Results | |
| ECE | $(769) million | $(416) million | $(63) million | $(492) million |
| Payout | 0 % | 100 % | 200 % | 79 % |
| 61 | 2024 Proxy Statement |  |
Consequently, the 20222023 annual cash incentive payments paid to the NEOs were as follows:
| | | | 2022 Base Salary ($) | | | 2022 Target Bonus (%) | | | 2022 Company Achievement (%) | | | 2022 Bonus Payout ($) | | |
| | Mr. Simmons | | | 1,125,000 | | | 130 | | | 93 | | | 1,360,125 | |
| | Mr. Young | | | 639,000 | | | 80 | | | 93 | | | 475,416 | |
| | Dr. de Brabander(1) | | | 667,819 | | | 70 | | | 93 | | | 434,750 | |
| | Dr. Cabral | | | 572,000 | | | 70 | | | 93 | | | 372,372 | |
| | Mr. Modi(2) | | | 525,000 | | | 70 | | | 93 | | | 274,356 | |
2023 Base ($) | 2023 Target Bonus (%) | 2023 Company Achievement (%) | 2023 Bonus Payout ($) | | |
| Mr. Simmons | 1,200,000 | 130 | 79 | 1,232,400 | |
| Mr. Young | 661,400 | 80 | 79 | 418,005 | |
| Dr. de Brabander(1) | 709,748 | 70 | 79 | 392,491 | |
| Dr. Cabral | 592,000 | 70 | 79 | 327,376 | |
| Mr. Bettington(2) | 620,000 | 70 | 79 | 269,591 | |
| (1) | Dr. de Brabander’s bonus payout was |
| (2) | Mr. |
Long-Term Incentive Awards
To further incentivize shareholder value creation, beginning in 2022, we changed the mix ofgrant equity awards granted to our executive leadership under our long-term incentive plan. In 2022,plan, consisting of the following mix changed from 75% performance awards and 25% RSUs toin 2023: 50% performance stock units,PAs, 25% RSUs and 25% stock options. The inclusion of stock options in 2023 rewards overperformance in relation toabsolute value creation and aligns our internal pay-for-performance strategy with continued equity appreciation for shareholders. This mix of equity awards is aligned with those of several companies in the peer group we use to benchmark our executive compensation program, as further described in “—Executive Compensation Process—Compensation Benchmarking” above.
The long-term incentive component of the NEOs’ compensation is designed to align this critical compensation element with our key financial incentives and focus leaders on achieving certain determined company performance objectives. In 2022,2023, NEOs received the following annual grant equity awards:
| ● | Performance Awards |
| ● | Restricted Stock Units (“RSUs”) (25% of total award opportunity): These awards have the potential to vest in roughly one-third increments on each of the first three annual anniversaries of the grant date, subject to continued employment with us. |
| ● | Stock Options (“Stock Options”) (25% of total award opportunity): These awards have the potential to vest in roughly one-third increments on each of the first three annual anniversaries of the grant date, subject to continued employment with us. |
The Compensation and Human Capital Committee has the discretion to adjust Performance AwardsPAs to take into account certain situations as set forth in the stock plan as follows:
| ● | the impact from the operations of any divestiture, such as a major product, business line or geography; |
| ● | the impact of any acquisitions, significant collaborations, restructuring or external litigation; |
| ● | foreign currency fluctuation impact greater than a 2% change to applicable plan rates; |
| ● | the impact of any non-GAAP adjustment provided each adjustment is approved by the committee; and/or |
| ● | any unforeseen adjustment provided such adjustment is approved by the committee. |
No dividends or dividend equivalents accrue on either the PAs or the RSUs.
| 62 | 2024 Proxy Statement |  |
2023 Target Grant Values
Our policy with respect to the annual equity award for all eligible employees, including the NEOs, is to grant the award and set the grant price at the Compensation and Human Capital Committee’scommittee’s regularly scheduled meeting during the first quarter of each year. For the 20222023 equity awards, we set the total target values for the NEOs based on internal pay equity, our company-wide performance, individual performance and our peer group data. As described in “Executive Compensation Process” above, we continue to work to better alignfollowing the completion of our executive compensationmulti-year “glidepath” approach, our NEOs' long-term incentive rewards (“LTIs”) are generally aligned with market practices to reflect the glidepath approach for each NEO.peer group medians. Total target values for the 20222023 equity grants to the NEOs were accordingly set as follows, the levels of which remained below the peer group medians:follows:
Equity Grant ($) | | ||||
Mr. Simmons | | ||||
Mr. Young | | ||||
Dr. de Brabander | | ||||
Dr. Cabral | | ||||
Mr. | |
| (1) | Mr. |
PERFORMANCE AWARDS
In March 2022,2023, each NEO (other than Mr. Bettington who joined the Company on March 20, 2023) received PAs under the Amended and Restated 2018 Elanco Animal Health Incorporated Stock Plan. These awards represent a substantial, at-risk component of NEO compensation directly tied to our long-term financial performance. The PAs granted in 20222023 have a two-year performance period from the beginning of 20222023 through the end of 20232024 and the final payout multiple per grant is determined by averaging the payout multiple of each of the two performance years. Due to the transformational nature of our 2018 initial public offering, our 2019 full separation from Eli Lilly, our former parent company, and our 2020 acquisition of Bayer Animal Health, the Compensation and Human Capital Committee believes it is appropriate to set the performance period at two years until a more consistent pattern of performance can be better established. The Compensation and Human Capital Committee regularly evaluates the two-year performance period to determine whether it continues to be appropriate. The Compensation and Human Capital Committee believes that this multi-year linkage will further reinforce a culture of long-term ownership and rewards year-over-year improvement and efficient use of capital and aligns award payouts with long-term shareholder returns.
The Compensation and Human Capital Committee approved Adjusted EBITDAR as the sole companyCompany performance measure for Elanco performance awards under our long-term incentive program. This measure was selected to replace Adjusted Net Income, which was used for Elanco performance awards in 2021 and 2020. Target Adjusted EBITDAR is defined as the prior year’s adjusted EBITDAR (Adjusted EBITDA plus R&D expense, excluding depreciation), plus the pre-tax required return on the year-over-year change in Gross Investments, as defined in the “2022“2023 Annual Cash Incentive” section above.
| Elanco Animal Health Incorporated | 63 | 2024 Proxy Statement |
The target number of PAs granted to an NEO was determined by dividing 50% of the target equity grant value applicable to such NEO by the closing stock price at the date of grant. The grant date fair values for these awards are reported in the table below.
Payout Date | Grant Date Fair Value ($)(1) | | ||||||
Mr. Simmons | | |||||||
Mr. Young | | |||||||
Dr. de Brabander | | |||||||
Dr. Cabral | | |||||||
Mr. | |
| (1) | All awards had a fair market value of |
| (2) | Mr. |
|  |
Payouts for the PAs granted in 20222023 range from 0% to 200% of target based on our achievement of the Target Adjusted EBITDAR and subject to continuous employment through the vesting date. Because the gross operating asset total used for the required return on investment is an average of the four quarters of the performance year, the Target Adjusted EBITDAR, which would result in a 1.0100% payout, cannot be determined until the end of a given performance year. In addition, each year’s target is set relative to the prior year’s results, so the 20232024 target is dependent on 20222023 EBITDAR performance. We believe that this required return on investment and multi-year linkage are key metric differentiators that encourage an efficient use of assets as well as a long-term ownership mindset. The final award payout is an average of Adjusted EBITDAR achieved, relative to Target, for each of the two performance years.
PAs Granted in 20212022 That Were Paid Based Upon Performance at December 31, 20212022 and 20222023
In 2021,2022, we granted PAs to the NEOs (other than Dr. de Brabander and Mr. Modi who were not with Elanco at that time) that were subject to a two-year performance cycle that ended on December 31, 2022.2023. The following describes the performance objectives, outcomes and shares earned under these 20212022 PA grants.
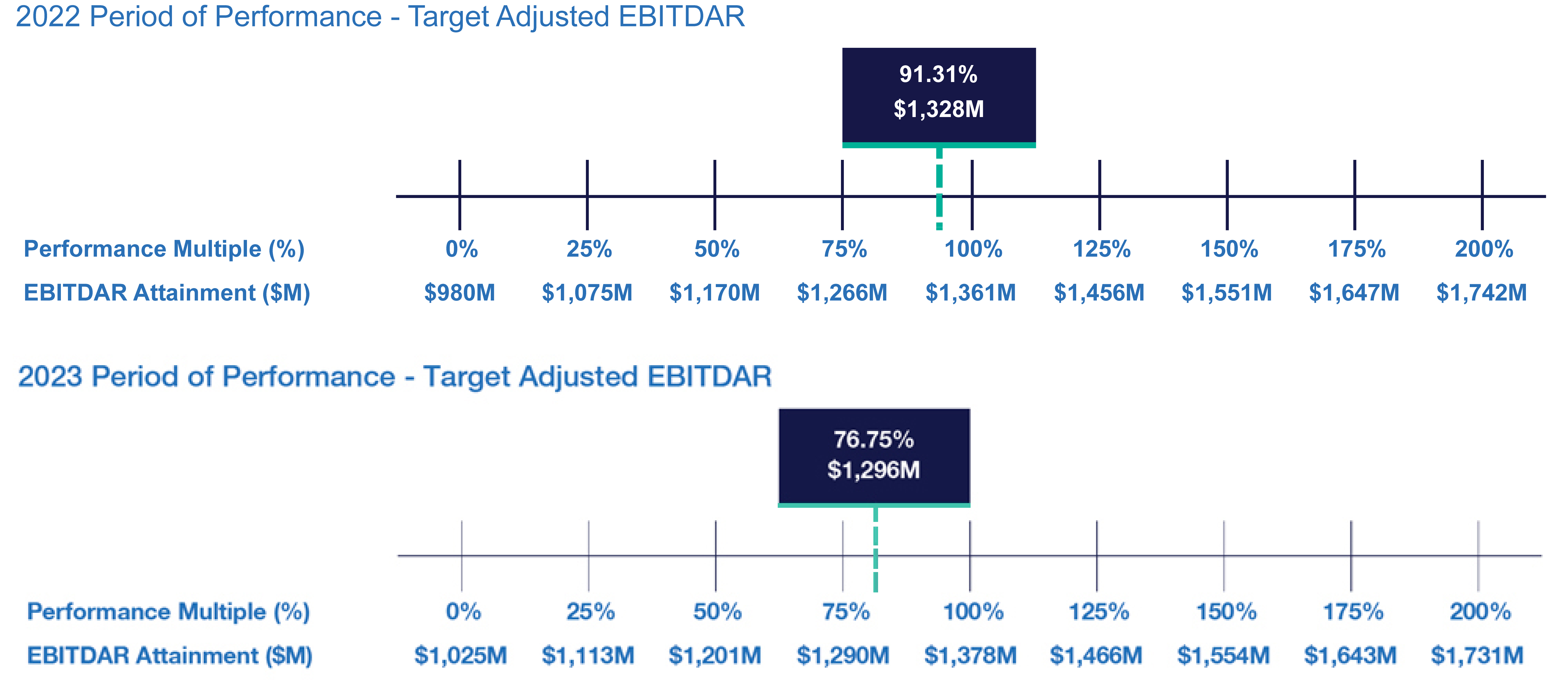
| Elanco Animal Health Incorporated | 64 | 2024 Proxy Statement |
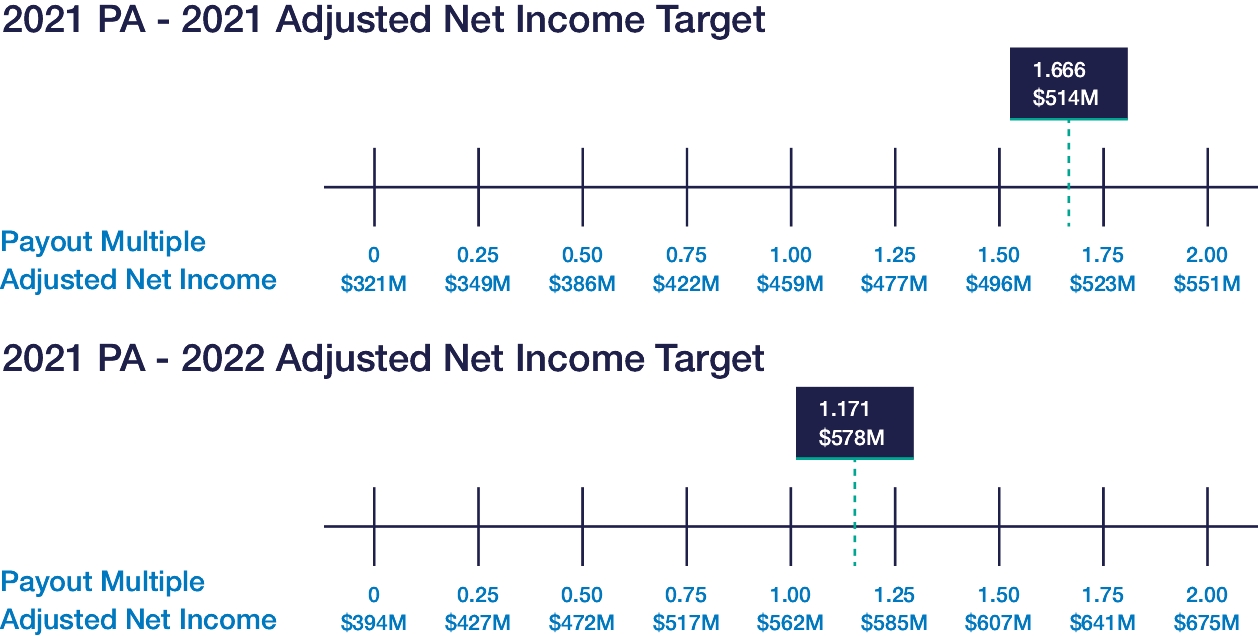
As Gross Investments is a key input on both the Compensation and Human Capital Committee was, and continuesAnnual Cash Incentive as well as the Performance Awards, the committee elected to be, based upon the Company’s original audited financials for the year ended December 31, 2021 even though those financials were revised in connection with the audit of the Company’s fiscal year ended December 31, 2022.continue using a standardized definition on Gross Investments across both programs. As indicatedpreviously discussed in the applicable plan,"2023 Annual Cash Incentive" section above, the awards based upon 2021 Adjusted Net Income had been previously certified and were not subjectCommittee made the decision to change absent a situation provided for inexclude several non-operational balance sheet items from the Company’s clawback policy. In any event, the revisions would have increased Adjusted Net Income to $522 million, which would have increased the PA award by approximately 4%.
The Committee also elected to apply the same normalization of capitalized R&D to account for pre-BAH acquisition expense as permitted bywas applied to the Annual Cash Incentive. Like the equivalent adjustment on ECE, normalizing the pre-BAH R&D expense required an adjustment to Gross Investment amounts for both 2022 and 2023, as the capital charge is applied to the year-over-year change in Gross Investments. As the Committee did not make this adjustment until the second half of 2023, it elected not to apply this same change to the 2022 performance period attainment.
An itemized breakdown of the changes to the Gross Investments definition, and their resulting impact on Target Adjusted EBITDAR and the Performance Award Agreements. We calculated this adjustment based onMultiple for the foreign currency rate impact on revenue that exceeded a threshold2023 period of +/- 2%, and then applied any offsetting savings driven by those same rate fluctuations. For 2022, this approach yielded a positive $33 million adjustment to Adjusted Net Income. The Compensation and Human Capital Committee believes that this approach is fair and balanced, as the committee would apply a downward adjustmentperformance, are set forth in the eventtable below. In aggregate, all changes decreased our Target Adjusted EBITDAR attainment by $6 million, lowering our 2023 attainment by 2%. Because our final payout is an average of a positive impact from foreign currency rates above 2% threshold. This approach adjusts for a material macroeconomic factorthe two performance years, and the Committee did not relatedmake any adjustments to core business performance, which aligns with our pay-for-performance compensation philosophy.2022, the Gross Investment changes reduced the final payout by 1%.
Gross Investments Definition Changes - Performance Award Impact
| 2023 Adjustment Category | EBITDAR Target Impact ($M) | 2023 PA Attainment Multiple Impact (%) | |
| FX and Impairments to Goodwill | (41) | (12) | |
| FX on Gross Intangibles | 19 | 5 | |
| Rate Changes on Accrued Pension | 6 | 2 | |
| Pre-BAH Normalized Capitalized R&D | 10 | 3 | |
| Total Adjustments | (6) | (2) |
For the NEOs who participatedreceived these awards in these 2021 awards,2022, the number of our shares earned under the performance-based awardsPAs is the average of the two payout multiples (1.666(91.31% for 20212022 and 1.17176.75% for 2022) times2023) multiplied by the target number of shares, andwhich is set forth in the table below. Details on the vesting schedule of these awards are set forth below in “Executive Compensation Tables—Outstanding Equity Awards at December 31, 2022.2023.”
| Name | Target Shares | Shares Paid Out | |
| Mr. Simmons | 176,227 | 148,084 | |
| Mr. Young | 43,366 | 36,440 | |
| Dr. de Brabander | 28,992 | 24,362 | |
| Dr. Cabral | 25,398 | 21,342 | |
| 65 | 2024 Proxy Statement |  |
| | Name | | | Target Shares | | | Shares Paid Out | |
| | Mr. Simmons | | | 223,121 | | | 316,386 | |
| | Mr. Young | | | 47,006 | | | 66,655 | |
| | Dr. Cabral | | | 28,507 | | | 40,423 | |
Restricted Stock Units
On March 1, 2022,2023, each NEO also received RSUs under the Amended and Restated 2018 Stock Plan. These time-based awards are designed to align the interests of the NEOs with the interests of our shareholders by promoting the retention of our NEOs over the longer term.
RSUs vest over a three-year period, with 33% of the award vesting on the first anniversary of the grant date, 33% of the award vesting on the second anniversary of the grant date and 34% of the award vesting on the third anniversary of the grant date, subject to continuous service through each vesting date. The number of RSUs granted to an NEO was determined by dividing 25% of the target equity grant value applicable to such NEO by the closing stock price at the date of grant. The grant date fair values for these awards are reported in the table below.
Date | Grant Date Fair Market Value ($)(1) | | ||||||
Mr. Simmons | | |||||||
Mr. Young | | |||||||
Dr. de Brabander | | |||||||
Dr. Cabral | ||||||||
Mr. | |
| (1) | All awards had a fair market value of |
| (2) | Mr. |
Stock Options
On March 1, 2022,2023, each NEO also received stock options under the 2018 Stock Plan. These time-based awards are designed to incentivize management to drive increases in stock price and align the interests of the NEOs with the interests of our shareholders.
The number of stock options each NEO receives is determined by dividing each person’s stock option equity target divided by the fair market value based on Black-Scholes modeling as of the grant date. Because the option exercise price is equivalent to the market close price on the grant date, the options require the stock to appreciate for our NEOs to realize any compensation. For the 20222023 grants, based on the option fair market valuation of $10.89$4.93 and the exercise price of $28.94,$11.26, our stock price would have to increase approximately 38%44% above the exercise price for our NEOs to realize the grant date fair market values shown in the table below.
Stock options vest over a three-year period, with 33% of the award vesting on the first anniversary of the grant date, 33% of the award vesting on the second anniversary of the grant date and 34% of the award vesting on the third anniversary of the grant date, subject to continuous service through each vesting date. The number of stock options granted to an NEO was determined by dividing 25% of the target equity grant value applicable to such NEO by the closing stock price atoption fair market value based on Black-Scholes modeling as of the date of grant.grant date. The grant date fair values for these awards are reported in the table below.
Date |  |
Grant Date Fair Market Value ($)(1) | | |||||||
Mr. Simmons | | |||||||
Mr. Young | | |||||||
Dr. de Brabander | | |||||||
Dr. Cabral | ||||||||
Mr. |
| (1) | All awards had a fair market value of |
| (2) | Mr. |
| Elanco Animal Health Incorporated | 66 | 2024 Proxy Statement |
SIGN-ON AWARDS AND SPECIAL PERFORMANCE AWARD
During 2023, the Committee approved the following sign-on and special performance awards:
| • | Sign-on Awards for Mr. Bettington – in connection with Mr. Bettington’s hire as our Executive Vice President, Global Strategy and Market Development in April 2023, the Committee approved the payment to Mr. Bettington of a $100,000 cash sign-on bonus that is subject to repayment as to a pro-rated amount of the bonus in the event that Mr. Bettington leaves Elanco during the first three years of employment for any reason other than an involuntary termination without cause. In lieu of other annual equity awards, Mr. Bettington received an RSU with a grant date fair value of $1,000,000 that vests as to one-half of the shares on April 3, 2024 and 2025. |
| • | Special Performance Award for Dr. de Brabander – A special performance-based stock award with a grant date fair value at target of $500,000 was issued to Dr. de Brabander in October 2023 to incentivize her efforts leading the work toward 2024 regulatory approvals for our pipeline products. The award will vest in February 2025, subject to achievement of critical R&D milestones, with payouts ranging from 70% to 130% of target. |
OTHER BENEFITS
Benefits are an important part of retention and financial security for all employees, and each of the benefits described below are designed to provide a market-competitive executive compensation program. In addition to the benefits described below, the NEOs are eligible to participate in our health and welfare programs, matching gifts program and other employee benefit programs on the same basis as other employees.
Elanco Executive Severance Plan
Each NEO is eligible to participate in the Elanco Executive Severance Pay Plan. This Plan, which defines the circumstances where an NEO is entitled to receive severance benefits in the event his or her employment with us is terminated. A description of the Elanco Executive Severance Plan and the benefits to which an NEO may be entitled is set forth in the narrative disclosure accompanying the table in “Executive Compensation Tables—Potential Payments Upon Termination or Change in Control (as of December 31, 2022)2023)” below.
2018 Change in Control Severance Pay Plan for Select Employees
Each NEO is eligible to participate in the Elanco 2018 Change in Control Severance Pay Plan for Select Employees, which provides severance benefits to an NEO in the event his or her employment with us is terminated following a change in control. A description of the Elanco 2018 Change in Control Severance Pay Plan for Select Employees and the benefits to which an NEO may be entitled is set forth in “Executive Compensation Tables — Payment Upon Termination or Change in Control (as of December 31, 2022)2023) — Elanco Change in Control Severance Pay Plan for Select Employees” below.
The Elanco 401(k) Plan
We provide retirement benefits to eligible employees, which includes our NEOs, through the Elanco 401(k) Plan, a defined contribution plan qualified under Sections 401(a) and 401(k) of the Code. Participants may elect to contribute a portion of their base salary to the plan, and we match employee contributions up to 6% of base salary (subject to IRS limits). In addition, in 2023, we provideprovided a non-elective contribution in the amount of 3% of base salary earnings, contingent on active employment on December 31 of each year.31. The employee contributions, our contributions and earnings thereon are paid out in accordance with elections made by the participant under the terms and conditions of the Elanco 401(k) Plan.
| Elanco Animal Health Incorporated | 67 | 2024 Proxy Statement |
The Elanco Deferred Compensation Program
The NEOs may defer receipt of all or part of their annual cash incentive bonus under the Elanco Deferred Compensation Plan, which allows participants to save for retirement in a tax-effective way at minimal cost to us. Under this unfunded and non-qualified plan, amounts deferred by the participant are credited at an interest rate of 120% of the applicable federal long-term rate, as described in more detail in “Executive Compensation Tables—Nonqualified Deferred Compensation” below. In 2022,Starting in 2024, the Compensation and Human Capital Committee approved new plan features for the Elanco Deferred Compensation Plan to both promote ownership and ensure that our offerings are in line with our peer group. The revised Deferred Compensation Plan will include company stock and 401(k)-type fund options for deferrals of base and bonus. Elections of employer stock will be eligible for a match on the first 6% of each NEO’s
 |
GOVERNANCE AND OTHER MATTERS
Employment Agreements
We do not have employment agreements with any of the NEOs. We do not provide excise tax payments, reimbursements, or gross-ups to any of the NEOs.
Stock Ownership and Holding Guidelines
Our Board has adopted stock ownership guidelines for our executive officers, which are designed to further promote long-term shareholder value creation and help ensure our senior executives remain focused on both short- and long-term objectives. The individuals have a period of five years from the date of starting in his/her current role to meet the guidelines. Shares of Elanco stock and outstanding RSU awards held by the individual are counted toward the requirement. PAs and stock options do not count toward the requirement. Mr. Simmons, our Chief Executive Officer, is required to own Elanco common stock with a value equal to at least six times his annual base salary. Each of the other NEOs is required to own Elanco common stock with a value equal to at least three times their annual base salary. All of the NEOs are required to hold at least 50% of all equity awards granted until their stock ownership requirements are satisfied, which allows them to build toward their respective ownership requirements. As of the Compensation and Human Capital Committee’s most-recent annual governance review in May 2022,2023, each of the NEOs was in compliance with the stock ownership guidelines or was making appropriate progress towards meeting the applicable ownership level within a reasonable period of time.
Anti-Hedging and Anti-Pledging Policy
We have a formal policy under which Non-Employee Directorsnon-employee directors and employees are not permitted to purchase financial instruments (including prepaid variable forward contracts, equity swaps, collars and exchange funds) or otherwise engage in transactions that hedge or offset any decrease in the market value of a company’s equity securities granted to the employee or director as compensation or held directly or indirectly by the employee or director. Additionally, our Corporate Governance Guidelines state that our directors are prohibited from hedging their Elanco stock and from pledging, or using as collateral, their Elanco stock.
Executive Compensation Recovery Policies
All of our incentive awards generally are subject to forfeiture upon termination of employment prior to the end of the performance or vesting period or for disciplinary reasons. The Committee adopted a Required Compensation Recovery Policy ("Required Policy") in compliance with SEC rules and NYSE listing standards that provides for the recoupment of Erroneously Awarded Compensation (as defined in the policy) during the covered period from the applicable former and current executive officers, including the NEOs, in the event we are required to prepare an accounting restatement due to material non-compliance with any financial reporting requirement. The Required Policy was filed as an exhibit to the Company's Annual Report for the year ended December 31, 2023. In addition, the Committee adopted a Supplemental Compensation and Human Capital Committee has adopted an executive compensation recovery policyRecovery Policy (the "Supplemental Policy") that gives the Compensation and Human Capital Committee broad discretion to claw back incentive payouts from any member of our senior management, which includes the NEOs, whosein the event of misconduct caused or contributed to Elanco having to restate all or a portionrestatement of its financial statements or resulted infinancials. Misconduct is defined as a material violation of law or policy that causes significant harm to Elanco (or(and includes an individual who failed in his or her supervisory responsibility to manage or monitor conduct or risks appropriately and such failure contributed materially to the harm caused to Elanco).
| Elanco Animal Health Incorporated | 68 | 2024 Proxy Statement |
The Supplemental Policy covers any incentive compensation awarded or paid to an employee at a time when he or she is a member of our senior management.management and, in the event of misconduct, includes any equity award and severance payments. Subsequent changes in status, including retirement or termination of employment, do not affect our rights to recover compensation under the policy. Recovery can extend back as far as three years.
Tax Deductibility of Compensation Expense
Section 162(m) of the Code generally places a $1 million limit on the amount of compensation a publicly held company can deduct in any tax year on compensation paid to “covered employees,” including the NEOs. While the Compensation and Human Capital Committee may consider tax deductibility as one of many factors in determining executive compensation, the Compensation and Human Capital Committee will award compensation that it determines is consistent with the goals of our executive compensation program even if such compensation is not tax deductible by us, if it determines that payment of such compensation is consistent with our business needs.
 |
COMPENSATION RISK OVERSIGHT
We monitor the risks associated with our compensation program and individual executive compensation decisions on an ongoing basis. The Compensation and Human Capital Committee, in collaboration with its independent compensation consultant, WTW, identified no material risks in our executive compensation programs in 2022.2023. In their 2022its 2023 annual risk analysis of our incentive compensation plans, WTW used certain evaluation criteria for incentive awards to determine whether or not these incentive plans were reasonably likely to incentivize risk-taking among those who participate in them, including, among others, the following:
| • | The metrics used to determine payout under the incentive plans; |
| • | Whether or not the metrics used to determine payout under the incentive plans were balanced; |
| • | The maximum incentive pay multiple; |
| • | The funding thresholds under the incentive plans; |
| • | The performance period for each plan; |
| • | The level of management that may exercise discretion as to the ultimate payout under the incentive plans; and |
| • | Any deferrals, holdbacks or clawback mechanisms under the incentive plans. |
The metrics used to determine payout under the incentive plans;
| Elanco Animal Health Incorporated | 69 | 2024 Proxy Statement |
Compensation and Human Capital Committee Report
To the extent that this Proxy Statement has been or will be specifically incorporated by reference into any other filing of Elanco under the Securities Act or the Exchange Act, this section entitled “Compensation and Human Capital Committee Report” will not be deemed to be so incorporated, unless specifically provided otherwise in such filing.
The Compensation and Human Capital Committee is primarily responsible for reviewing, approving and overseeing Elanco’s compensation plans and practices and works with management and the Committee’scommittee’s independent compensation consultant to establish Elanco’s compensation philosophy and programs. The Compensation and Human Capital Committee has reviewed and discussed the “Compensation Discussion and Analysis” section of this Proxy Statement with management. Based on this review and discussion, the Compensation and Human Capital Committee recommended to the Board of Directors that the “Compensation Discussion and Analysis” section be included in Elanco’s 20222023 Annual Report on Form 10-K for the year ended December 31, 20222023 (incorporated by reference) and in this Proxy Statement.
Respectfully submitted,
Lawrence E. Kurzius (Chair)
R. David Hoover
Kirk P. McDonald
Denise Scots-Knight
| 70 | 2024 Proxy Statement |  |
Executive | Tables |
The following table summarizes compensation awarded to, earned by and/or paid to our NEOs in connection with their service to Elanco during 2023, 2022 2021 and 2020,2021, as applicable.
Name and Principal Position | | | Year | | | Salary ($) | | | Bonus ($)(4) | | | Stock Awards ($)(5) | | | Option Awards ($)(6) | | | Non-Equity Incentive Plan Compensation ($)(7) | | | All Other Compensation ($)(8) | | | Total Compensation ($) |
Jeffrey Simmons President and CEO | | | 2022 | | | 1,125,000 | | | — | | | 7,650,029 | | | 2,550,002 | | | 1,360,125 | | | 28,476 | | | 12,713,632 |
| | 2021 | | | 1,046,000 | | | — | | | 9,261,019 | | | — | | | 1,725,900 | | | 27,096 | | | 12,060,015 | ||
| | 2020 | | | 1,025,000 | | | — | | | 6,860,048 | | | — | | | 1,045,500 | | | 28,272 | | | 8,958,820 | ||
Todd Young Executive Vice President and Chief Financial Officer | | | 2022 | | | 639,000 | | | — | | | 1,882,518 | | | 627,504 | | | 475,416 | | | 30,046 | | | 3,654,484 |
| | 2021 | | | 580,000 | | | — | | | 2,109,014 | | | — | | | 574,200 | | | 28,696 | | | 3,291,910 | ||
| | 2020 | | | 568,000 | | | — | | | 1,649,020 | | | — | | | 337,960 | | | 28,960 | | | 2,583,940 | ||
Ellen de Brabander(1) Executive Vice President, Innovation, Regulatory, and Business Development | | | 2022 | | | 667,819 | | | — | | | 1,258,543 | | | 419,505 | | | 434,750 | | | 29,426 | | | 2,810,043 |
Ramiro Cabral(2) Executive Vice President, Elanco International | | | 2022 | | | 572,000 | | | — | | | 1,102,527 | | | 367,505 | | | 372,372 | | | 28,446 | | | 2,442,850 |
| | 2021 | | | 475,000 | | | — | | | 1,279,037 | | | — | | | 438,900 | | | 27,209 | | | 2,220,146 | ||
Bobby Modi(3) Executive Vice President, U.S. Pet Health and Global Digital Transformation | | | 2022 | | | 419,602 | | | 600,000 | | | 900,017 | | | — | | | 274,356 | | | 28,446 | | | 2,222,421 |
Summary Compensation Table
| Name and Principal Position | Year | Salary ($) | Bonus ($) | Stock Awards ($)(3) | Option Awards ($)(4) | Non-Equity Incentive Plan Compensation ($)(5) | All Other Compensation ($)(6) | Total ($) | |
| Jeffrey Simmons President and CEO | 2023 | 1,200,000 | — | 8,400,005 | 2,800,003 | 1,232,400 | 30,761 | 13,663,169 | |
| 2022 | 1,125,000 | — | 7,650,029 | 2,550,002 | 1,360,125 | 28,476 | 12,713,632 | ||
| 2021 | 1,046,000 | — | 9,261,019 | — | 1,725,900 | 27,096 | 12,060,015 | ||
| Todd Young Executive Vice President and Chief Financial Officer | 2023 | 661,400 | — | 1,961,267 | 653,753 | 418,005 | 32,296 | 3,726,720 | |
| 2022 | 639,000 | — | 1,882,518 | 627,504 | 475,416 | 30,046 | 3,654,484 | ||
| 2021 | 580,000 | — | 2,109,014 | — | 574,200 | 28,696 | 3,291,910 | ||
| Ellen de Brabander(1) Executive Vice President, Innovation and Regulatory | 2023 | 709,748 | — | 2,031,513 | 510,502 | 392,491 | 39,480 | 3,683,732 | |
| 2022 | 667,819 | — | 1,258,543 | 419,505 | 434,750 | 29,426 | 2,810,043 | ||
| Ramiro Cabral Executive Vice President, President International | 2023 | 592,000 | — | 1,275,015 | 425,001 | 327,376 | 31,362 | 2,650,754 | |
| 2022 | 572,000 | — | 1,102,527 | 367,505 | 372,372 | 28,446 | 2,442,850 | ||
| 2021 | 475,000 | — | 1,279,037 | — | 438,900 | 27,209 | 2,220,146 | ||
| Timothy Bettington(2) Executive Vice President, Global Strategy and Market Development | 2023 | 486,528 | 100,000 | 1,000,008 | — | 269,591 | 197,835 | 2,053,962 |
| (1) | Dr. de Brabander |
| (2) |
| Mr. |
| (3) |
| This column shows the grant date fair value of the RSUs and PAs awarded to the NEOs in |
| 71 | 2024 Proxy Statement |  |
The table below shows the target and maximum payouts for the 20222023 PAs included in this column of the “Summary Compensation Table.”
Name | | | Payout Date | | | Target Payout ($) | | | Maximum Payout ($) |
Mr. Simmons | | | February 2023 | | | 5,100,009 | | | 10,200,019 |
Mr. Young | | | February 2023 | | | 1,255,012 | | | 2,510,024 |
Dr. de Brabander | | | February 2023 | | | 839,028 | | | 1,678,057 |
Dr. Cabral | | | February 2023 | | | 735,018 | | | 1,470,036 |
Mr. Modi | | | February 2023 | | | 0 | | | 0 |
| Name | Payout Date | Target Payout ($) | Maximum Payout ($) | |
| Mr. Simmons | February 2025 | 5,600,003 | 11,200,007 | |
| Mr. Young | February 2025 | 1,307,511 | 2,615,022 | |
| Dr. de Brabander | February 2025 | 1,021,001 | 2,042,001 | |
| Dr. Cabral | February 2025 | 850,006 | 1,700,012 | |
| Mr. Bettington | February 2025 | — | — |
| (4) | This column includes on-cycle Elanco Non-Qualified Stock Options, granted on March 1, |
| (5) | This column shows payments under the Elanco Corporate Bonus Plan (the “Elanco Bonus Plan”) for performance in 2023, 2022 |
| (6) | The amounts in this column for Messrs. Simmons |
| Elanco Animal Health Incorporated | 72 | 2024 Proxy Statement |
Grants of Plan Based Awards
The following table summarizes the grants of plan-based awards to the NEOs during 20222023 under each of the following plans: the Elanco Bonus Plan (a non-equity incentive plan) and the Amended and Restated 2018 Stock Plan, which provides for the grant of PAs, RSUs and stock options. To receive a payout under the PAs, RSUs or to vest in stock options, a participant must remain employed with Elanco through the end of the relevant performance period or vesting date (except in the case of death, disability, retirement, or redundancy). No dividends or dividend equivalents, if any were to be declared and paid, would accrue on either the PAs or the RSUs.
| | | | | | Elanco Compensation and Human Capital Committee | | | Estimated Future Payouts Under Non-Equity Incentive Plan Awards(1) | | | Estimated Future Payouts Under Equity Incentive Plan Awards | | | All Other Stock Awards: Number of Shares of Stock or Units (#) | | | All Other Option Awards: Number of Securities Underlying Options (#) | | | Exercise or Base Price of Option Awards ($) | | | Grant Date Fair Value of Stock and Option Awards ($) | | |||||||||||
| | Name | | | Award | | | Grant Date(2) | | | Approval Date | | | Target ($) | | | Maximum ($) | | | Target (#) | | | Maximum (#) | | ||||||||||||
| | Mr. Simmons | | | 2022 Elanco Bonus Plan | | | — | | | — | | | 1,462,500 | | | 2,925,000 | | | | | | | | | | | | | | ||||||
| | 2022 PAs(3) | | | 3/1/2022 | | | 2/22/2022 | | | | | | | 176,227 | | | 352,454 | | | | | | | | | 5,100,009 | | ||||||||
| | 2022 RSUs(4) | | | 3/1/2022 | | | 2/22/2022 | | | | | | | | | | | 88,114 | | | | | | | 2,550,019 | | |||||||||
| | 2022 Elanco Options(5) | | | 3/1/2022 | | | 2/22/2022 | | | | | | | | | | | | | 234,160 | | | 28.94 | | | 2,550,002 | | ||||||||
| | Mr. Young | | | 2022 Elanco Bonus Plan | | | — | | | — | | | 511,000 | | | 1,022,400 | | | | | | | | | | | | | | ||||||
| | 2022 PAs(3) | | | 3/1/2022 | | | 2/21/2022 | | | | | | | 43,366 | | | 86,732 | | | | | | | | | 1,255,012 | | ||||||||
| | 2022 RSUs(4) | | | 3/1/2022 | | | 2/21/2022 | | | | | | | | | | | 21,683 | | | | | | | 627,506 | | |||||||||
| | 2022 Elanco Options(5) | | | 3/1/2022 | | | 2/21/2022 | | | | | | | | | | | | | 57,622 | | | 28.94 | | | 627,504 | | ||||||||
| | Dr. de Brabander | | | 2022 Elanco Bonus Plan(8) | | | — | | | — | | | 467,473 | | | 934,947 | | | | | | | | | | | | | | ||||||
| | 2022 PAs(3) | | | 3/1/2022 | | | 2/21/2022 | | | | | | | 28,992 | | | 57,984 | | | | | | | | | 839,028 | | ||||||||
| | 2022 RSUs(4) | | | 3/1/2022 | | | 2/21/2022 | | | | | | | | | | | 14,496 | | | | | | | 419,514 | | |||||||||
| | 2022 Elanco Options(5) | | | 3/1/2022 | | | 2/21/2022 | | | | | | | | | | | | | 38,522 | | | 28.94 | | | 419,505 | | ||||||||
| | Dr. Cabral | | | 2022 Elanco Bonus Plan | | | — | | | — | | | 400,400 | | | 800,800 | | | | | | | | | | | | | | ||||||
| | 2022 PAs(3) | | | 3/1/2022 | | | 2/21/2022 | | | | | | | 25,398 | | | 50,796 | | | | | | | | | 735,018 | | ||||||||
| | 2022 RSUs(4) | | | 3/1/2022 | | | 2/21/2022 | | | | | | | | | | | 12,699 | | | | | | | 367,509 | | |||||||||
| | 2022 Elanco Options(5) | | | 3/1/2022 | | | 2/21/2022 | | | | | | | | | | | | | 33,747 | | | 28.94 | | | 367,505 | | ||||||||
| | Mr. Modi | | | 2022 Elanco Bonus Plan(9) | | | — | | | — | | | 295,007 | | | 590,014 | | | | | | | | | | | | | | ||||||
| | 2022 Sign-On RSU Award(6) | | | 4/1/2022 | | | 2/21/2022 | | | | | | | | | | | 34,457 | | | | | | | 900,017 | | |||||||||
| Elanco Compensation and Human Capital Committee | Estimated Future Payouts Under Non-Equity Incentive Plan Awards(1) | Estimated Future Payouts Under Equity Incentive Plan Awards | All Other Stock Awards: Number of Shares of Stock or Units (#) | All Other Option Awards: Number of Securities Underlying Options (#) | Exercise or Base Price of Option Awards ($) | Grant Date Fair Value of Stock and Option Awards ($)(6) | | ||||||
| Name | Award | Grant Date(2) | Approval Date | Target ($) | Maximum ($) | Threshold (#) | Target (#) | Maximum (#) | |||||
| Mr. Simmons | 2023 Elanco Bonus Plan | — | — | 1,560,000 | 3,120,000 | | |||||||
| 2023 PAs(3) | 3/1/23 | 2/24/23 | — | 497,336 | 994,672 | 5,600,003 | |||||||
| 2023 RSUs(4) | 3/1/23 | 2/24/23 | 248,668 | 2,800,002 | | ||||||||
| 2023 Elanco Options(5) | 3/1/23 | 2/24/23 | 567,952 | 11.26 | 2,800,003 | | |||||||
| Mr. Young | 2023 Elanco Bonus Plan | — | — | 529,120 | 1,058,240 | | |||||||
| 2023 PAs(3) | 3/1/23 | 2/23/23 | — | 116,120 | 232,240 | 1,307,511 | | ||||||
| 2023 RSUs(4) | 3/1/23 | 2/23/23 | 58,060 | 653,756 | | ||||||||
| 2023 Elanco Options(5) | 3/1/23 | 2/23/23 | 132,607 | 11.26 | 653,753 | ||||||||
| Dr. de Brabander | 2023 Elanco Bonus Plan(7) | — | — | 496,824 | 993,647 | | |||||||
| 2023 PAs(3) | 3/1/23 | 2/23/23 | — | 90,675 | 181,350 | 1,021,001 | |||||||
| 2023 RSUs(4) | 3/1/23 | 2/23/23 | 45,338 | 510,506 | |||||||||
| 2023 Elanco Options(5) | 3/1/23 | 2/23/23 | 103,550 | 11.26 | 510,502 | ||||||||
| 2023 R&D Off-Cycle PA (9) | 10/2/23 | 8/15/23 | 31,935 | 45,621 | 59,308 | 500,006 | |||||||
| Dr. Cabral | 2023 Elanco Bonus Plan | — | — | 414,400 | 828,800 | | |||||||
| 2023 PAs(3) | 3/1/23 | 2/23/23 | — | 75,489 | 150,978 | 850,006 | |||||||
| 2023 RSUs(4) | 3/1/23 | 2/23/23 | 37,745 | 425,009 | | ||||||||
| 2023 Elanco Options(5) | 3/1/23 | 2/23/23 | 86,207 | 11.26 | 425,001 | ||||||||
| Mr. Bettington | 2023 Elanco Bonus Plan(8) | — | — | 341,255 | 682,510 | ||||||||
| 2023 Sign-On RSU Award(10) | 4/3/23 | 2/17/23 | 108,109 | 1,000,008 | | ||||||||
 |
| Target and maximum payouts for performance under the Elanco Bonus Plan. Bonus payouts range from 0% to 200% of target. The target and maximum amounts represent ECE attainments of |
| (2) | The annual grant date for Elanco awards is established in advance of the grant date by the |
| (3) | Represents the range of payouts for |
| 73 | 2024 Proxy Statement |
| (4) | Represents the shares underlying the |
| (5) | Represents the shares underlying the |
| (6) |
| Represents the grant date fair value of the equity awards granted in |
| (7) | Amounts denominated in Euros have been converted to USD using the average daily rate of €1 to |
| (8) | Represents target and maximum payouts based upon pro-rated eligible earnings. |
| (9) | Represents the range of payouts for a 2023 R&D Off-Cycle PA. This PA will pay out in February 2025, with payouts ranging from 70% to 130% of target. The grant date fair value of the PAs is based on the probable payout outcome at the time of grant. The grant date fair value assuming payout at target and maximum are listed for these awards in Note 3 to the “Summary Compensation Table” above. |
| (10) | Represents the shares underlying the 2023 sign-on RSU award for Mr. Bettington when he joined Elanco. Half of the shares underlying this grant vested on April 3, 2024, and the remainder will vest on April 3, 2025. |
| Elanco Animal Health Incorporated | 74 | 2024 Proxy Statement |  |
Outstanding Equity Awards at December 31, 20222023
The closing price of our common stock on December 31, 2022,2023, which was $12.22,$14.90, was used to calculate the values in the table below.
| Option Awards | Stock Awards | |||||||||
| Name | Award | Number of Securities Underlying Options Exercisable (#) | Number of Securities Underlying Options Unexercisable (#) | Option Exercise Price ($) | Option Expiration Date | Number of Shares or Units of Stock That Have Not Vested (#) | Market Value of Shares or Units of Stock That Have Not Vested ($) | Equity Incentive Plan Awards: Number of Unearned Shares, Units, or Other Rights That Have Not Vested (#) | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units, or Other Rights That Have Not Vested ($) | |
| Mr. Simmons | 2018 Stock Options(1) | 109,642 | — | 31.61 | 10/20/2028 | |||||
| 2021 RSUs(2) | 25,288 | 376,791 | ||||||||
| 2022 - 2023 Executive PAs(3) | 176,227 | 2,625,782 | ||||||||
| 2022 RSUs(4) | 59,037 | 879,651 | ||||||||
| 2022 Elanco Options(5) | 77,272 | 156,888 | 28.94 | 3/1/2032 | ||||||
| 2023 - 2024 Executive PAs(6) | 497,336 | 7,410,306 | ||||||||
| 2023 RSUs(7) | 248,668 | 3,705,153 | ||||||||
| 2023 Elanco Options(8) | — | 567,952 | 11.26 | 3/1/2033 | ||||||
| Mr. Young | 2021 RSUs(2) | 5,329 | 79,402 | |||||||
| 2022 - 2023 Executive PAs(3) | 43,366 | 646,153 | ||||||||
| 2022 RSUs(4) | 14,528 | 216,467 | ||||||||
| 2022 Elanco Options(5) | 19,015 | 38,607 | 28.94 | 3/1/2032 | ||||||
| 2023 - 2024 Executive PAs(6) | 116,120 | 1,730,188 | ||||||||
| 2023 RSUs(7) | 58,060 | 865,094 | ||||||||
| 2023 Elanco Options(8) | — | 132,607 | 11.26 | 3/1/2033 | ||||||
| Dr. de Brabander | 2022 - 2023 Executive PAs(3) | 28,992 | 431,981 | |||||||
| 2022 RSUs(4) | 9,713 | 144,724 | ||||||||
| 2022 Elanco Options(5) | 12,712 | 25,810 | 28.94 | 3/1/2032 | ||||||
| 2023 - 2024 Executive PAs(6) | 90,675 | 1,351,058 | ||||||||
| 2023 RSUs(7) | 45,338 | 675,536 | ||||||||
| 2023 Elanco Options(8) | — | 103,550 | 11.26 | 3/1/2033 | ||||||
| 2023 R&D Off-Cycle PA(9) | 45,621 | 679,753 | ||||||||
| Elanco Animal Health Incorporated | 75 | 2024 Proxy Statement |
| | | | | | Option Awards | | | Stock Awards | | ||||||||||||||||||||
| | Name | | | Award | | | Number of Securities Underlying Options Exercisable (#) | | | Number of Securities Underlying Options Unexercisable (#) | | | Option Exercise Price ($) | | | Option Expiration Date | | | Number of Shares or Units of Stock That Have Not Vested (#) | | | Market Value of Shares or Units of Stock That Have Not Vested ($) | | | Equity Incentive Plan Awards: Number of Unearned Shares, Units, or Other Rights That Have Not Vested (#) | | | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units, or Other Rights That Have Not Vested ($) | |
| | Mr. Simmons | | | 2018 Stock Options(1) | | | 109,642 | | | | | 31.61 | | | 10/20/2028 | | | | | | | | | | |||||
| | 2020 RSUs(2) | | | | | | | | | | | 20,992 | | | 256,522 | | | | | | |||||||||
| | 2021 - 2022 Executive PAs(3) | | | | | | | | | | | | | | | 223,121 | | | 2,726,539 | | |||||||||
| | 2021 RSUs(4) | | | | | | | | | | | 49,831 | | | 608,935 | | | | | | |||||||||
| | 2022 - 2023 Executive PAs(5) | | | | | | | | | | | | | | | 176,227 | | | 2,153,494 | | |||||||||
| | 2022 RSUs(6) | | | | | | | | | | | 88,114 | | | 1,076,753 | | | | | | |||||||||
| | 2022 Elanco Options(7) | | | | | 234,160 | | | 28.94 | | | 3/1/2032 | | | | | | | | | | ||||||||
| | Mr. Young | | | 2020 RSUs(2) | | | | | | | | | | | 5,046 | | | 61,662 | | | | | | ||||||
| | 2021 - 2022 Executive PAs(3) | | | | | | | | | | | | | | | 47,006 | | | 574,413 | | |||||||||
| | 2021 RSUs(4) | | | | | | | | | | | 10,499 | | | 128,298 | | | | | | |||||||||
| | 2022 - 2023 Executive PAs(5) | | | | | | | | | | | | | | | 43,366 | | | 529,933 | | |||||||||
| | 2022 RSUs(6) | | | | | | | | | | | 21,683 | | | 264,966 | | | | | | |||||||||
| | 2022 Stock Options(7) | | | | | 57,622 | | | 28.94 | | | 3/1/2032 | | | | | | | | | | ||||||||
| | Dr. de Brabander | | | 2021 Sign-On RSU Award(8) | | | | | | | | | | | 22,818 | | | 278,836 | | | | | | ||||||
| | 2022 - 2023 Executive PAs(5) | | | | | | | | | | | | | | | 28,992 | | | 354,282 | | |||||||||
| | 2022 RSUs(6) | | | | | | | | | | | 14,496 | | | 177,141 | | | | | | |||||||||
| | 2022 Stock Options(7) | | | | | 38,522 | | | 28.94 | | | 3/1/2032 | | | | | | | | | | ||||||||
| | Dr. Cabral | | | 2018 Stock Options(1) | | | 21,086 | | | | | 31.61 | | | 10/20/2028 | | | | | | | | | | |||||
| | 2020 RSUs(2) | | | | | | | | | | | 2,956 | | | 36,122 | | | | | | |||||||||
| | 2021 - 2022 Executive PAs(3) | | | | | | | | | | | | | | | 28,507 | | | 348,356 | | |||||||||
| | 2021 RSUs(4) | | | | | | | | | | | 6,368 | | | 77,817 | | | | | | |||||||||
| | 2022 - 2023 Executive PAs(5) | | | | | | | | | | | | | | | 25,398 | | | 310,364 | | |||||||||
| | 2022 RSUs(6) | | | | | | | | | | | 12,699 | | | 155,182 | | | | | | |||||||||
| | 2022 Stock Options(7) | | | | | 33,747 | | | 28.94 | | | 3/1/2032 | | | | | | | | | | ||||||||
| | Mr. Modi | | | 2022 Sign-On RSU Award(9) | | | | | | | | | | | 34,457 | | | 421,065 | | | | | | ||||||
| Option Awards | Stock Awards | |||||||||
| Name | Award | Number of Securities Underlying Options Exercisable (#) | Number of Securities Underlying Options Unexercisable (#) | Option Exercise Price ($) | Option Expiration Date | Number of Shares or Units of Stock That Have Not Vested (#) | Market Value of Shares or Units of Stock That Have Not Vested ($) | Equity Incentive Plan Awards: Number of Unearned Shares, Units, or Other Rights That Have Not Vested (#) | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units, or Other Rights That Have Not Vested ($) | |
| Dr. Cabral | 2018 Stock Options(1) | 21,086 | — | 31.61 | 10/20/2028 | |||||
| 2021 RSUs(2) | 3,233 | 48,172 | ||||||||
| 2022 - 2023 Executive PAs(3) | 25,398 | 378,430 | ||||||||
| 2022 RSUs(4) | 8,509 | 126,784 | ||||||||
| 2022 Elanco Options(5) | 11,136 | 22,611 | 28.94 | 3/1/2032 | ||||||
| 2023 - 2024 Executive PAs(6) | 75,489 | 1,124,786 | ||||||||
| 2023 RSUs(7) | 37,745 | 562,401 | ||||||||
| 2023 Elanco Options(8) | — | 86,207 | 11.26 | 3/1/2033 | ||||||
| Mr. Bettington | 2023 Sign-On RSUs(10) | 108,109 | 1,610,824 | |||||||
| (1) | An award of nonqualified stock options granted after our 2018 initial public offering, which vested on October 20, 2021 and has a seven-year exercise period ending October 20, 2028. |
| (2) |
| RSUs granted on March 1, 2021. One-third of the shares underlying this grant vested on March 1, 2022, one-third vested on March 1, 2023, and the remainder |
| (3) | PAs granted for the 2022-2023 performance period, to the extent earned, |
| (4) | RSUs granted on March 1, 2022. One-third of the shares underlying this grant vested on March 1, 2023, one-third |
 |
| Elanco nonqualified stock options granted on March 1, 2022. One-third of the options underlying this grant vested on March 1, 2023, one-third |
| (6) | PAs ranted for the 2023-2024 performance period, to the extent earned, vested following the close of the performance period. |
| (7) | RSUs granted on March 1, 2023. One-third of the shares underlying this grant vested on March 1, 2024, one-third will vest on March 1, 2025, and the remainder will vest on March 1, 2026. |
| (8) | Elanco nonqualified stock options granted on March 1, 2023. One-third of the options underlying this grant vested on March 1, 2024, one-third will vest on March 1, 2025, and the remainder will vest on March 1, 2026. The grant expires March 1, 2033, ten years after the grant date. |
| (9) | This award was granted to Dr. de Brabander as part of |
| (10) | This award was granted to Mr. |
| 76 |
Stock Vested in 2023
Stock Awards | | |||||||
Number of Shares Acquired on Vesting (#) | | Value Realized on Vesting ($)(1) | | |||||
| | ||||||||
47,177(3) | | 521,100 | | |||||
| Dr. de Brabander | | | ||||||
Dr. Cabral | ||||||||
— | ||||||||
| (1) | Amounts reflect the market value of Elanco’s common stock on the date of vesting. |
| (2) | For Mr. Simmons, this represents |
| (3) | For Mr. Young, this represents |
| (4) | For Dr. de Brabander, this represents |
| (5) | For Dr. Cabral this represents |
| (6) | Mr. |
 |
| | Name | | | Executive Contributions in Last Fiscal Year ($) | | | Aggregate Earnings in Last Fiscal Year ($) | | | Aggregate Balance at Last Fiscal Year End ($)(1) | |
| | Dr. Cabral | | | — | | | 10,351 | | | 579,302 | |
Nonqualified Deferred Compensation
| Name | Executive Contributions in Last Fiscal Year ($) | Aggregate Earnings in Last Fiscal Year ($) | Aggregate Withdrawals/ Distributions ($) | Aggregate Balance at Last Fiscal Year End ($)(1) | |
| Dr. Cabral | — | 26,436 | (66,430) | 539,308 | |
The “Nonqualified Deferred Compensation” table above shows information about the Elanco Deferred Compensation Plan. Our executives may defer receipt of all or part of their cash bonus under the Elanco Deferred Compensation Plan. Of the NEOs, only Dr. Cabral participates in the Elanco Deferred Compensation Plan. Amounts deferred by executives under the plan are credited with interest at 120% of the applicable federal long-term rate as established the preceding December by the U.S. Treasury Department under Section 1274(d) of the Code, with monthly compounding. Deferral elections for bonuses earned in 2022,2023, payable in 2023,2024, were made in the fourth quarter of 2021.2022. Participants may elect to receive the funds in a lump sum or in up to ten annual installments following termination of employment, butor may elect in-service distributions payable in a lump sum or in up to five annual installments as early as the second year after the end of the plan year in which they were deferred. Once a distribution schedule is elected, it may not make withdrawals while employed by us,be cancelled or accelerated, except in the event of hardship as approved by the Compensation and Human Capital Committee.committee. All deferral elections and associated distribution schedules are irrevocable. The Elanco Deferred Compensation Plan is unfunded and amounts deferred under the plan are subject to forfeiture in the event of our bankruptcy.
| 77 | 2024 Proxy Statement |  |
In the fourth quarter of 2023, Elanco opened enrollment for an enhanced and expanded Non-Qualified Deferred Compensation Plan, which will apply to compensation earned in 2024. We elected to augment our existing plan to both ensure that our executive benefits offerings were competitive relative to the broader market and to further reinforce our ownership culture. Additional plan features include expanded deferral sources, increased investment options, and a company stock match enhancement. Participants had the option to defer 2024 compensation of up to 70% of their base pay, 100% of their bonus, and any full-value equity awards. Base and bonus deferrals were eligible for expanded investment options, including a notional 401(k)-style fund menu and notional Elanco stock. Deferrals made into Elanco stock will receive a 1:1 company match up to 6% of a participant’s base and bonus pay. We believe that this company match enhancement, which cliff vests two years after the end of a given plan year, aligns the interests of our senior leaders with shareholders while also helping to retain key talent.
Payments Upon Termination or Change in Control (As of December 31, 2022)2023)
The following table describes the potential payments and benefits under our compensation and benefit plans and arrangements to which the applicable NEOs would have been entitled upon a hypothetical termination of employment on December 31, 2022,2023, in the circumstances described in the table. The closing price of our common stock on December 31, 2022,2023, which was $12.22,$14.90, was used to calculate the values in the table below. The narrative following the tabular disclosure below contains more detail on the treatment of certain equity awards upon a qualifying termination (defined as a termination due to death, disability, site or plant closing, or restructuring) of employment for the NEOs. Other than the payments and benefits described below, any agreement to provide severance payments or benefits would be at the discretion of the Compensation and Human Capital Committee.committee. These severance benefits are contingent upon each executing a release of claims in favor of Elanco and agreeing to certain other customary post-employment covenants, except in the case of death.
| | | | Cash Severance Payment(1) ($) | | | Continuation of Medical / Welfare Benefits (present value) ($) | | | Value of Acceleration of Equity Awards ($) | | | Total Termination Benefits ($) | | |
| | Mr. Simmons | | | | | | | | | | ||||
| | • Termination due to death | | | — | | | — | | | 5,745,502(3) | | | 5,745,502 | |
| | • Termination due to disability, reduction in force, or other qualified reason not in connection with change in control | | | 5,175,000 | | | 46,164(5) | | | 4,561,194(3) | | | 9,782,358 | |
| | • Non-qualified discharge not in connection with change in control | | | 5,175,000 | | | 46,164(5) | | | —(6) | | | 5,221,164 | |
| | • Change in control | | | 5,175,000 | | | 36,567(2) | | | 6,822,243(4) | | | 12,033,809 | |
| | Mr. Young | | | | | | | | | | ||||
| | • Termination due to death | | | — | | | — | | | 1,294,306(3) | | | 1,294,306 | |
| | • Termination due to disability, reduction in force, or other qualified reason not in connection with change in control | | | 1,150,200 | | | 24,396(5) | | | 1,016,606(3) | | | 2,191,202 | |
| | • Non-qualified discharge not in connection with change in control | | | 1,150,200 | | | 24,396(5) | | | —(6) | | | 1,174,596 | |
| | • Change in control | | | 2,300,400 | | | 38,617(2) | | | 1,559,272(4) | | | 3,898,289 | |
| | Dr. de Brabander(7) | | | | | | | | | | ||||
| | • Termination due to death | | | — | | | — | | | 633,118(3) | | | 633,118 | |
| | • Termination due to disability, reduction in force, or other qualified reason not in connection with change in control | | | 1,135,293 | | | — | | | 271,822(3) | | | 1,407,114 | |
| | • Non-qualified discharge not in connection with change in control | | | 1,135,293 | | | — | | | —(6) | | | 1,135,293 | |
| | • Change in control | | | 2,270,586 | | | 4,808(2) | | | 810,259(4) | | | 3,085,653 | |
| | Dr. Cabral | | | | | | | | | | ||||
| | • Termination due to death | | | — | | | — | | | 772,658(3) | | | 772,658 | |
| | • Termination due to disability, reduction in force, or other qualified reason not in connection with change in control | | | 972,400 | | | 23,134(5) | | | 608,434(3) | | | 1,603,967 | |
| | • Non-qualified discharge not in connection with change in control | | | 972,400 | | | 23,134(5) | | | —(6) | | | 995,534 | |
| | • Change in control | | | 1,944,800 | | | 36,649(2) | | | 927,840(4) | | | 2,909,289 | |
| | Mr. Modi | | | | | | | | | | ||||
| | • Termination due to death | | | — | | | — | | | 421,065(3) | | | 421,065 | |
| | • Termination due to disability, reduction in force, or other qualified reason not in connection with change in control | | | 892,500 | | | — | | | 158,041(3) | | | 1,050,541 | |
| | • Non-qualified discharge not in connection with change in control | | | 892,500 | | | — | | | —(6) | | | 892,500 | |
| | • Change in control | | | 1,785,000 | | | 2,023(2) | | | 421,065(4) | | | 2,208,088 | |
| 78 | 2024 Proxy Statement |
| Cash Severance Payment(1) ($) | Continuation of Medical / Welfare Benefits (present value) ($) | Value of Acceleration of Equity Awards ($) | Total Termination Benefits ($) | | |
| Mr. Simmons | | ||||
| Termination due to death | — | — | 13,359,876(2) | 13,359,876 | |
| Termination due to disability, reduction in force, or other qualified reason not in connection with change in control | 5,520,000 | 65,810(3) | 8,589,295(2) | 14,175,105 | |
| Non-qualified discharge not in connection with change in control | 5,520,000 | 65,810(3) | —(4) | 5,585,810 | |
| Qualifying termination in connection with change in control | 5,520,000 | 51,302(5) | 17,065,030(6) | 22,636,331 | |
| Mr. Young | | ||||
| Termination due to death | — | — | 3,154,900(2) | 3,154,900 | |
| Termination due to disability, reduction in force, or other qualified reason not in connection with change in control | 1,190,520 | 32,324(3) | 2,036,900(2) | 3,259,744 | |
| Non-qualified discharge not in connection with change in control | 1,190,520 | 32,324(3) | —(4) | 1,222,844 | |
| Qualifying termination in connection with change in control | 2,381,040 | 50,510(5) | 4,019,994(6) | 6,451,544 | |
| Dr. de Brabander(7) | | ||||
| Termination due to death | — | — | 2,644,583(2) | 2,644,583 | |
| Termination due to disability, reduction in force, or other qualified reason not in connection with change in control | 1,206,571 | — | 1,796,203(2) | 3,002,775 | |
| Non-qualified discharge not in connection with change in control | 1,206,571 | — | —(4) | 1,206,571 | |
| Qualifying termination in connection with change in control | 2,413,143 | 5,452(5) | 3,659,973(6) | 6,078,568 | |
| Dr. Cabral | | ||||
| Termination due to death | — | — | 1,991,980(2) | 1,991,980 | |
| Termination due to disability, reduction in force, or other qualified reason not in connection with change in control | 1,006,400 | 33,317(3) | 1,273,965(2) | 2,313,682 | |
| Non-qualified discharge not in connection with change in control | 1,006,400 | 33,317(3) | —(4) | 1,039,717 | |
| Qualifying termination in connection with change in control | 2,012,800 | 51,999(5) | 2,554,366(6) | 4,619,165 | |
| Mr. Bettington | | ||||
| Termination due to death | — | — | 1,610,824(2) | 1,610,824 | |
| Termination due to disability, reduction in force, or other qualified reason not in connection with change in control | 1,054,000 | 33,317(3) | 598,563(2) | 1,685,880 | |
| Non-qualified discharge not in connection with change in control | 1,054,000 | 33,317(3) | —(4) | 1,087,317 | |
| Qualifying termination in connection with change in control | 2,108,000 | 51,999(5) | 1,610,824(6) | 3,770,823 | |
| (1) | As of December 31, |
| (2) | For Mr. Simmons, the amount includes 2021 RSUs, 2022 – 2023 PAs, 2022 RSUs, 2022 stock options, 2023 – 2024 PAs, 2023 RSUs, and 2023 stock options. For Mr. Young, the amount includes 2021 RSUs, 2022 – 2023 PAs, 2022 RSUs, 2022 stock options, 2023 - 2024 PAs, 2023 RSUs, and 2023 stock options. For Dr. de Brabander, the amount includes 2022 – 2023 PAs, 2022 RSUs, 2022 stock options, 2023 - 2024 PAs, 2023 RSUs, and 2023 stock options. For Dr. Cabral, the amount includes 2021 RSUs, 2022 – 2023 PAs, 2022 RSUs, 2022 stock options, 2023 – 2024 PAs, 2023 RSUs, and 2023 stock options. For Mr. Bettington, the amount includes 2023 sign-on RSUs. The value of all PAs are based on target level of performance. |
| (3) | See “Elanco Executive Severance Plan” below for a discussion of payments following a termination not related to a change in control. |
| (4) | Termination due to performance is not considered a qualifying termination under the 2018 Stock Plan and the applicable grant agreements. |
| (5) | See “Elanco Executive Change in Control Severance Pay Plan for Select Employees” below for a discussion of payments following a change in control. |
 |
| Includes the acceleration of RSUs, PAs, stock options and sign-on RSUs, as applicable, upon the event of certain qualifying terminations following a change in control. The value of all PAs are based on target level of performance. |
| Amounts paid in Euros have been converted to USD using the average daily rate of €1 to |
| Elanco Animal Health Incorporated | 79 | 2024 Proxy Statement |
Equity Award Acceleration in Connection with a Change in Control or Certain Terminations of Employment
Upon a change in control of Elanco, unvested RSUs will continue to vest and pay out upon the earlier of (i) the completion of the original award period, (ii) upon a covered termination of employment as described below, or (iii) if the successor entity does not assume, substitute or otherwise replace the award, upon the change in control, as dictated by the RSU award agreement. PAs will be deemed earned at target upon a change in control, as dictated by the Performance AwardPA agreement.
Upon a qualified retirement and certain other terminations without cause, unvested equity awards will vest on a pro-rata basis.
Elanco Change in Control Severance Pay Plan for Select Employees
In connection with our initial public offering, our Board adopted change in control severance pay plans for nearly all Elanco employees, including a plan that applies to the NEOs. These severance pay plans are intended to preserve employee morale and productivity and encourage retention in the face of the disruptive impact of an actual or rumored change in control. In addition, these severance pay plans are intended to align our participating employees’ and our shareholders’ interests by enabling our executives to evaluate corporate transactions that may be in the best interests of our shareholders without undue concern over whether the transactions would jeopardize the participating employee’s own employment.
The basic elements of the select plan applicable to the NEOs include:
| • | Double trigger. Unlike “single trigger” plans that pay out immediately upon a change in control, the select plan requires a “double trigger” — a change in control followed by an involuntary loss of employment within two years. This is consistent with our intent to provide employees with financial protection resulting from a loss of employment. |
| • | Covered terminations.Our participating NEOs are eligible for payments under our severance pay plan if, within two years of the change in control, their employment is terminated (i) without “cause” by Elanco; or (ii) for “good reason” (e.g., a relocation or material reduction in title, work responsibilities, salary, variable pay potential, or benefits coverage) by the employee, each as is defined in the plan. |
| • | Severance payment. NEOs are eligible for two years’ base salary plus two times their target bonus for the then-current year. |
| • | Benefit continuation. Basic employee benefits such as health and life insurance would continue for a period of 18 months following a participating NEO’s termination of employment unless he or she becomes eligible for coverage with a new employer during that 18-month period. |
| • | No gross ups.In some circumstances, the payments or other benefits received by a participating employee in connection with a change in control could exceed limits established under Section 280G of the Code resulting in an excise tax payment. We would not reimburse or gross up employees for these taxes. However, the amount of benefits related to a change in control would be reduced to the maximum amount that would not result in an excise tax if the effect would be to deliver a greater after-tax benefit than the employee would receive if his or her benefits were not so reduced. |
| Elanco Animal Health Incorporated | 80 | 2024 Proxy Statement |
Elanco Executive Severance Pay Plan
In November 2020, we adopted the Elanco Executive Severance Pay Plan for our senior employees, including the NEOs. We adopted this plan following the Compensation and Human Capital Committee’scommittee’s discussions with its independent compensation consultant, WTW, to fill a gap in our compensation programs and align them with market practices. Under the plan, severance benefits are payable to eligible employees if their employment is terminated by us without cause and in certain other specified circumstances that are not in connection with a change in control. The plan does not provide for benefits upon voluntary separation of service by the employee.
 |
The severance benefits provided under the plan are as follows:
| • | A lump sum severance payment equal to the sum of (i) two times the amount of base salary for the CEO, or one times the base salary for other executives; plus (ii) two times (with respect to the CEO) or one times (with respect to other executives) the amount of their target annual cash incentive bonus for the year of termination or, if there is no target-based annual cash incentive bonus, then the annual cash bonus paid or payable for the most recently completed calendar year; plus (iii) a lump sum payment equal to 24 months (with respect to CEO) or 12 months (with respect to other executives) of Elanco contributions paid for active employees for medical and dental coverage. |
| • | Outplacement services for up to 12 months following the termination date. |
CEO Pay Ratio
We are providing the following information about the relationship of the annual total compensation of our employees and the annual total compensation of our President and CEO, Jeffrey Simmons.
We have identified our median employee, using our employee population on December 31, 2022,2023, by use of a “consistently applied compensation measure” or “CACM.” We chose a CACM that closely approximates the annual total direct compensation of our non-contingent employees. Specifically, we identified the median employee by looking at annual base pay, bonus opportunity at target and the grant date fair value for standard equity awards. We then identified the median paid employee and calculated his or her total annual compensation in accordance with the requirements of the “Summary Compensation Table” above. We used a newthe same median employee in 2023 as we did in 2022, due to material changesbecause we believe there has not been any change in our employee population because ofor employment compensation arrangements that we believe would significantly alter our restructuring announced in December of 2021.pay ratio calculation.
In applying the CACM, we did not perform adjustments to the compensation paid to part-time employees to calculate what they would have been paid on a full-time basis and we chose not to include one-time equity awards when choosing the median employee, since the grant of such awards is not a recurring event. We also chose not to exclude any employees when determining our median employee.
For 2022,2023, the annual total compensation of our median employee was $59,388.$67,459. Mr. Simmons’ annual total compensation for 2022,2023, as reported in the “Summary Compensation Table” above, was $12,713,632.$13,663,169. The ratio of Mr. Simmons’ total compensation to the median employee’s total compensation was 214:203:1.
| 81 | 2024 Proxy Statement |  |
Pay Versus Performance |
Pay Versus Performance Tables
As required by the Dodd-Frank Wall Street Reform, Consumer Protection Act and Item 402(v) of Regulation S-K, we are providing the following information about the relationship between executive officer “compensation actually paid” (“CAP”) and certain Company financial performance metrics. CAP is calculated in accordance with Item 402(v) of Regulation S-K and differs from compensation shown in the Summary Compensation Table (“SCT”) on page 71. See below for a reconciliation of the total compensation shown in the SCT to CAP. The Compensation and Human Capital Committee makes executive compensation decisions independent of SEC disclosure requirements and reviews a variety of Company-wide and individual factors to link CAP to our Company and executive performance. For further information concerning our variable pay-for-performance philosophy and how we align executive compensation with our performance, refer to the “Compensation Discussion and Analysis” above.
| | | | | | | | | | | | Value of Initial Fixed $100 Investment Based On: | | | | | | ||||||||||
| | Year | | | Summary Compensation Table Total for CEO ($)(1)(2) | | | Compensation Actually Paid to CEO ($)(1)(2) | | | Average Summary Compensation Table Total for non-CEO NEOs ($)(1)(2) | | | Average Compensation Actually Paid to non-CEO NEOs ($)(1)(2) | | | Total Shareholder Return ($)(3) | | | Peer Group Total Shareholder Return ($)(3)(4) | | | Net Income (Loss) ($) | | | Adjusted Net Income (5) ($) | |
| | 2022 | | | 12,713,632 | | | (2,017,094) | | | 2,782,450 | | | 863,887 | | | 41 | | | 126 | | | (78,000,000) | | | 544,000,000 | |
| | 2021 | | | 12,060,015 | | | 16,525,957 | | | 3,202,470 | | | 3,753,652 | | | 96 | | | 149 | | | (483,000,000) | | | 522,000,000 | |
| | 2020 | | | 8,958,820 | | | 9,320,037 | | | 2,151,684 | | | 2,175,973 | | | 104 | | | 132 | | | (574,000,000) | | | 199,000,000 | |
Average Summary Comp. Table Total for non-CEO NEOs ($)(1)(2) | Average Comp. Actually Paid to non-CEO NEOs ($)(1)(2) | Value of Initial Fixed $100 Investment Based On:
| ||||||
| Year | Summary Comp. Table ($)(1)(2) | Comp. Actually Paid to ($)(1)(2) | Total Shareholder Return ($)(3) | S&P 500 ($)(3)(4) | Net Income (Loss) ($) | Adjusted ($)(5) | ||
| 2023 | 13,663,169 | 17,319,899 | 3,028,792 | 3,573,716 | 51 | 137 | (1,231,000,000) | 979,000,000 |
| 2022 | 12,713,632 | (2,017,094) | 2,782,450 | 863,887 | 41 | 136 | (78,000,000) | 1,017,000,000 |
| 2021 | 12,060,015 | 16,525,957 | 3,202,470 | 3,753,652 | 96 | 126 | (483,000,000) | 1,059,000,000 |
| 2020 | 8,958,820 | 9,320,037 | 2,151,684 | 2,175,973 | 104 | 108 | (574,000,000) | 529,000,000 |
| (1) | For |
For 2022, the CEO was our Chief Executive Officer Mr. Jeffrey Simmons, and the other NEOs were Mr. Todd Young- Executive Vice President and Chief Financial Officer; Dr. Ellen de Brabander- Executive Vice President, Innovation, Regulatory, and Business Development; Dr. Ramiro Cabral- Executive Vice President, Elanco International; and Mr. Bobby Modi, Executive Vice President, U.S. Pet Health and Global Digital Transformation.
For 2021, the CEO was our Chief Executive Officer Mr. Jeffrey Simmons, and the other NEOs were Mr. Todd Young- Executive Vice President and Chief Financial Officer; Mr. Aaron Schacht- Former Executive Vice President, Innovation, Regulatory, and Business Development; Ms. Joyce Lee- Former Executive Vice President, U.S. Pet Health and Commercial Operations; and Dr. Ramiro Cabral- Executive Vice President, Elanco International.
For 2020, the CEO was our Chief Executive Officer Mr. Jeffrey Simmons, and the other NEOs were Mr. Todd Young- Executive Vice President and Chief Financial Officer; Mr. Aaron Schacht- Former Executive Vice President, Innovation, Regulatory, and Business Development; Sarena Lin- Former Executive Vice President, Transformation and Technology; and Michael Bryant-Hicks- Former Executive Vice President, General Counsel and Corporate Secretary.
| 82 | 2024 Proxy Statement |
| (2) | A reconciliation of Total Compensation from the |
| | | | 2022 | | | 2021 | | | 2020 | | ||||||||||
| | Adjustments(a) | | | CEO ($) | | | Average of Other NEOs ($) | | | CEO ($) | | | Average of Other NEOs ($) | | | CEO ($) | | | Average of Other NEOs ($) | |
| | Total Compensation from SCT | | | 12,713,632 | | | 2,782,450 | | | 12,060,015 | | | 3,202,470 | | | 8,958,820 | | | 2,151,684 | |
| | Adjustments for stock and option awards: | | | | | | | | | | | | | | ||||||
| | Deduct: Grant date fair value of awards granted during fiscal year | | | (10,200,031) | | | (1,639,529) | | | (9,261,019) | | | (1,597,526) | | | (6,860,048) | | | (1,255,531) | |
| | Add: Fair value at year-end of awards granted during the covered fiscal year that are outstanding and unvested at year-end | | | 3,414,070 | | | 578,725 | | | 12,234,618 | | | 1,952,416 | | | 7,022,449 | | | 1,285,249 | |
| | Add: Year-over-year change in fair value of awards granted in prior fiscal year(s) that are outstanding and unvested at year end | | | (7,402,134) | | | (722,112) | | | 1,267,444 | | | 177,458 | | | 19,891 | | | (10,569) | |
| | Add: Change as of the vesting date (from the end of the prior fiscal year) in fair value of awards granted in any prior fiscal year for which vesting conditions were satisfied during such year | | | (542,631) | | | (135,647) | | | 224,899 | | | 18,833 | | | 178,925 | | | 5,139 | |
| | Compensation Actually Paid (as calculated) | | | (2,017,094) | | | 863,887 | | | 16,525,957 | | | 3,753,652 | | | 9,320,037 | | | 2,175,973 | |
| 2023 | ||
| Adjustments(a) | CEO ($) |
Average of Other NEOs ($) |
| Total Compensation from SCT | 13,663,169 | 3,028,792 |
| Adjustments for stock and option awards: | ||
| Deduct: SCT amount of Stock and Option awards | (11,200,008) | (1,964,264) |
| Add: Fair value at year-end of awards granted during the covered fiscal year that are outstanding and unvested at year-end | 14,502,230 | 2,460,549 |
| Add/Deduct: The difference between Year-over-year change in fair value of awards from the end of the granted in prior fiscal year(s) to the end of the applicable fiscal year that are outstanding and unvested at year end | 845,183 | 113,545 |
| Add: Change in fair value as of the vesting date (from the end of the prior fiscal year to the vesting date) in fair value of awards granted in any prior fiscal year for which vesting conditions were satisfied during such year | (490,675) | (64,905) |
| Compensation Actually Paid (as calculated) | 17,319,899 | 3,573,716 |
| (a) | All equity awards used to determine compensation actually paid to NEOs were re-valued according to FASB ASC Topic 718 and use |
 |
| Total shareholder return (“TSR”) is calculated based on a fixed investment of one hundred dollars measured from the market close on December 31, 2019 (the last trading day of 2019) through and including the end of the fiscal year for each year reported in the table. |
| (4) | For 2023, we have elected to begin using our 10-K peer group of the S&P 500 Pharmaceuticals Index to provide additional clarity and consistency across our external reporting. The peer group used |
For 2020,2023, the peer group consists of Agilent Technologies, Inc.; Alexion Pharmaceuticals, Inc.; BioMarin Pharmaceutical Inc.; Bio-Rad Laboratories, Inc.; DENTSPLY SIRONA Inc.; Edwards Lifesciences Corporation; Endo International plc; Hologic, Inc.; IDEXX Laboratories, Inc.; Incyte Corporation; Jazz Pharmaceuticals plc; Mettler-Toledo International Inc.; PerkinElmer, Inc.; Perrigo Company plc; STERIS plc; United Therapeutics Corporation; Varian Medical Systems, Inc.; West Pharmaceutical Services, Inc.; and Zoetis Inc.
For 2022, the peer group consistsconsisted of Agilent Technologies, Inc.; Alexion Pharmaceuticals, Inc.; Baxter International Inc.; Boston Scientific Corporation; Charles River Laboratories International, Inc.; DENTSPLY SIRONA Inc.; Edwards Lifesciences Corporation; Endo International plc; Hologic, Inc.; IDEXX Laboratories, Inc.; Incyte Corporation; Jazz Pharmaceuticals plc; Perrigo Company plc; Regeneron Pharmaceuticals, Inc.; STERIS plc; Varian Medical Systems, Inc.; Vertex Pharmaceuticals, Inc.; Zimmer Biomet Holdings, Inc.; and Zoetis Inc.
For 2021, the 2020 peer group; the return for 2021 reflects the return of the 2021 peer group consisted of Agilent Technologies, Inc.; Alexion Pharmaceuticals, Inc.; Baxter International Inc.; Boston Scientific Corporation; Charles River Laboratories International, Inc.; DENTSPLY SIRONA Inc.; Edwards Lifesciences Corporation; Endo International plc; Hologic, Inc.; IDEXX Laboratories, Inc.; Incyte Corporation; Jazz Pharmaceuticals plc; Perrigo Company plc; Regeneron Pharmaceuticals, Inc.; STERIS plc; Varian Medical Systems, Inc.; Zimmer Biomet Holdings, Inc.; and Zoetis Inc.
For 2020, the return for 2022 reflects the returnpeer group consisted of the 2022 peer group. Agilent Technologies, Inc.; Alexion Pharmaceuticals, Inc.; BioMarin Pharmaceutical Inc.; Bio-Rad Laboratories, Inc.; DENTSPLY SIRONA Inc.; Edwards Lifesciences Corporation; Endo International plc; Hologic, Inc.; IDEXX Laboratories, Inc.; Incyte Corporation; Jazz Pharmaceuticals plc; Mettler-Toledo International Inc.; PerkinElmer, Inc.; Perrigo Company plc; STERIS plc; United Therapeutics Corporation; Varian Medical Systems, Inc.; West Pharmaceutical Services, Inc.; and Zoetis Inc.
Returns for the 2020 peer group over the 3-yearfour-year period would have been $132, $156, $113, and $113,$120 for 2020, 2021, 2022, and 2022,2023, respectively. Returns for the 2021 peer group over the 3-yearfour-year-year period would have been $118, $133, $106, and $106,$119 for 2020, 2021, 2022, and 2022,2023, respectively. Returns for the 2022 peer group over the 3-yearfour-year-year period would have been $115, $139, $117, and $117,$136 for 2020, 2021, 2022, and 2023, respectively. Returns for the 2023 peer group over the four-year-year period would have been $125, $151, $117, and $133 for 2020, 2021, 2022, and 2023, respectively.
| 83 | 2024 Proxy Statement |
A comparison of our TSR, the TSR of our executive benchmarking peer group adjusted for each respective year's group, and the S&P 500 Pharmaceuticals TSR is below:
| TSR | ELAN ($) | Executive Benchmarking Peer Group ($) | S&P 500 Pharmaceuticals ($) |
| 2023 | 51 | 142 | 137 |
| 2022 | 41 | 126 | 136 |
| 2021 | 96 | 149 | 126 |
| 2020 | 104 | 132 | 108 |
| (5) | For |
Relationship between Pay and Performance
As described in the “Compensation Discussion and Analysis,” our executive compensation program reflects a pay-for-performance philosophy. While we utilize several performance measures to align executive compensation with Company performance, all of those Company measures are not presented in the Pay Versus Performance table above. Moreover, the Compensation and Human Capital Committee generally seeks to incentivize long-term performance, and therefore, does not specifically align the Company’s performance measures with CAP (as computed in accordance with Item 402(v) of Regulation S-K) for a particular year. Furthermore, both total compensation and CAP are impacted by various factors described in the “Compensation Discussion and Analysis,” including adjustments to NEO compensation over recent years on the glidepath to market competitive compensation.
In accordance with Item 402(v) of Regulation S-K, we are providing the following descriptions of the relationships between CAP and the information presented in the Pay Versus Performance table.
| Elanco Animal Health Incorporated | 84 | 2024 Proxy Statement |
CAP AND TSR versus Peer GroupVERSUS S&P 500 PHARMACEUTICALS INDEX
The chart below compares our TSR, peer groupS&P 500 Pharmaceuticals Index TSR, and CAP to our CEO and other NEOs (averaged) for the three-yearfour-year period from 2020 to 2022.2023. The graph compares the return on Elanco’s common stock with that of our market cap-weighted peer group used for executive compensation benchmarking from 2020, 2021, and 2022, respectively,the S&P 500 Pharmaceuticals Index, as described in footnote 4 from our Pay Versus Performance Table. The graph assumes a person invested $100 at market close on December 31, 2019 (the last trading day of 2019) in both Elanco and the relevant year’s peer group,S&P 500 Pharmaceuticals Index, and measures TSR, through and including the end of the fiscal year, for each year shown below, adjusted for each year’s respective peer group.below. This TSR calculation includes both stock price and dividends. It assumes that dividends paid by a company are reinvested in that company’s stock.

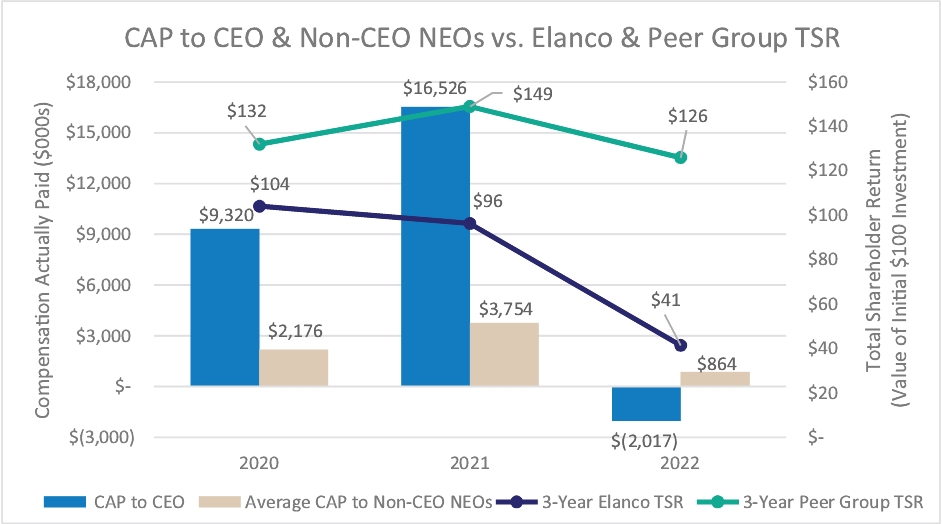
CAP AND GAAP NET INCOME (LOSS)
|  |
Executive pay is not linked to GAAP Net Income; therefore, there is a limited relationship between this metric and compensation actually paid.CAP.
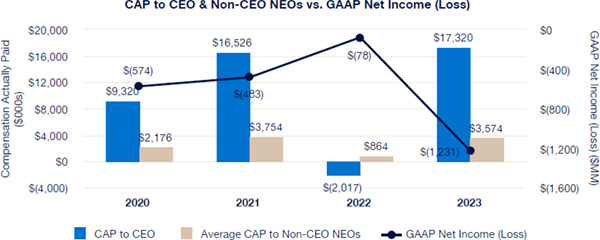
| Elanco Animal Health Incorporated | 85 | 2024 Proxy Statement |
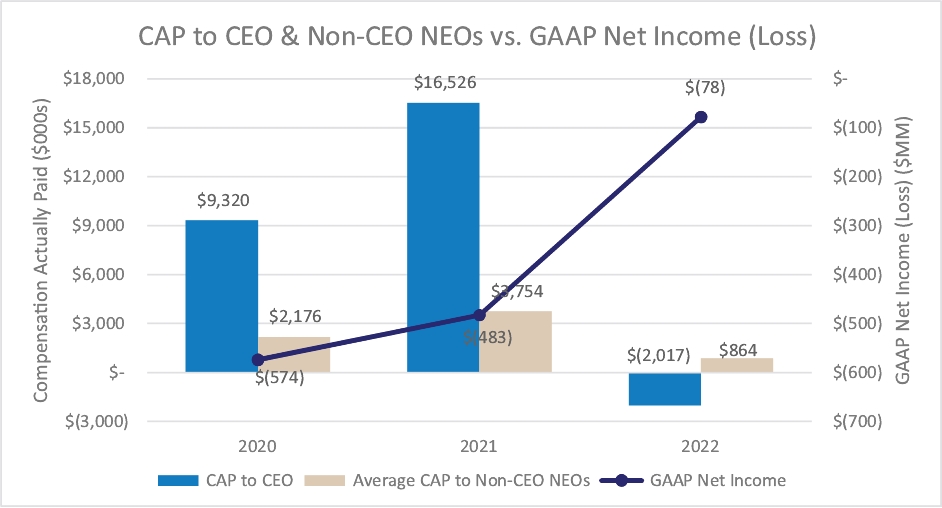
CAP and Adjusted Net Income (Loss)AND ADJUSTED EBITDA
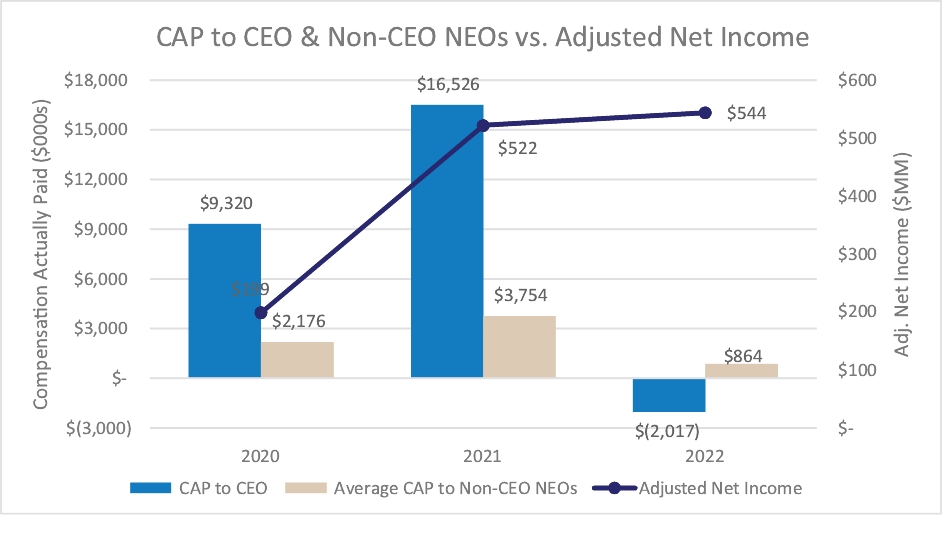
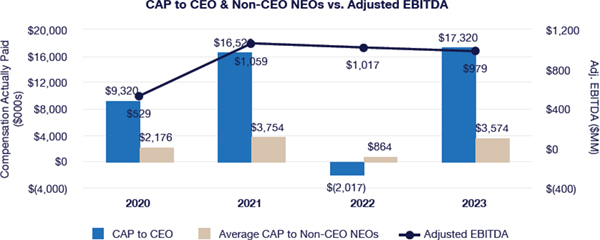
TABULAR LIST OF IMPORTANT FINANCIAL PERFORMANCE MEASURES
The following table lists the most important financial measures the Company used to link compensation actually paid to the NEOs for fiscal year 20222023 to our performance:
| Total Shareholder Return | |||||
Elanco Cash Earnings | |||||
| 86 | 2024 Proxy Statement |  |
Proposal No. |
Under our shareholders to approve the adoptioncurrent Articles of the AmendedIncorporation and Restated Elanco Animal Health Incorporated Employee Stock Purchase Plan (the “ESPP”). The ESPP was initially approved by shareholders at the 2022 Annual Meeting, and the first offering period commenced in late 2022 for employees of Elanco domestic subsidiaries only. The Compensation and Human Capital Committee andBylaws, our Board believe thatof Directors is divided into three classes, with the ESPP encourages our employees to acquire sharesterm of our common stock, fostering an ownership mentality among our employees and aligning their interests with thoseoffice of our shareholders, and will help us attract, retain and motivate talent in an increasingly competitive employment market.one class expiring at each annual meeting.
If shareholders approve this proposal, the amended and restated ESPP as presented in this Proxy Statement will become effective as of that date. If the amended and restated ESPPProposal No. 4 is not approved by our shareholders, the proposed amendments to the Articles of Incorporation would commence declassification of our Board at our 2025 annual meeting of shareholders (the “2025 Annual Meeting”). Directors elected at this Annual Meeting will be elected to three-year terms expiring at the annual meeting of shareholders held in 2027. If the proposed amendments are approved at this Annual Meeting, then, beginning with the ESPPclass of directors standing for election at the 2025 Annual Meeting, directors will not have an increased share reservebe elected to one-year terms of office. Directors currently serving terms that expire at the annual meetings of shareholders to be held in 2025 and it2026 will not(subject to their earlier resignation or removal) serve the remainder of their respective terms, and thereafter their successors will be offeredelected to employees outsideone-year terms. From and after the U.S.
With respect to Proposal No. 4, the proposed amendments to the Articles of Incorporation are included with this proxy statement as amended and restated, which we believe supports the approval of an expanded plan to provide competitive employee compensation opportunities.Appendix B. This summary does not purport to be a complete description of the ESPP andproposed amendments to the Articles of Incorporation is qualified in its entirety by reference to the full text of the amendments as set forth in Appendix B.
In proposing these amendments to the Articles of Incorporation and seeking to evolve our governance structure, our Board considered shareholder feedback and evolving governance practices. Our Board unanimously concluded, on the recommendation of the Corporate Governance Committee, that the proposed changes contemplated by this Proposal No. 4 should be made.
If this Proposal No. 4 is approved by the requisite vote of our shareholders at the Annual Meeting, the proposed amendments to the Articles of Incorporation would become effective upon the filing of amended and restated plan,Articles of Incorporation with the Secretary of State of the State of Indiana, which we would file promptly following the Annual Meeting if our shareholders approve the amendments. If this Proposal No. 4 is included as Appendix B to this Proxy Statement.
The ESPP Administrator may delegate its duties under the ESPP to Elanco employees or to outside firms, banks or other financial institutions. All questionsaffirmative vote of interpretation or applicationat least a majority of all of the ESPP are determined in the sole discretionoutstanding shares of the ESPP Administrator, and its decisions are final, conclusive, and binding upon all persons.
| 87 | 2024 Proxy Statement |  |
WhileAs noted below, we are also submitting proposals to amend our Articles of Incorporation to: change the ESPP Administrator has discretionvoting standard for uncontested director elections from a plurality standard to establish different offering periods, shares generally will be offereda majority standard (Proposal No. 5), enable shareholders to amend our Bylaws (Proposal No. 6), and enable shareholders to call special meetings of shareholders under the ESPP through consecutive offering periodscertain circumstances (Proposal No. 7). Approval of approximately three months that generally begin with the first business daythis Proposal No. 4 is not contingent on approval or disapproval of each fiscal quarter of each year. To participate in the ESPP, eligible employees must authorize payroll deductions in whole dollar amounts up to ten percent of base salary. Once an eligible employee becomes a participant in the ESPP, the employee may affirmatively elect to participate automatically in each successive offering period until such time as the employee withdraws from,Proposals No. 5, 6, or is no longer eligible to participate in, the ESPP.7.
The purchase price per share of our common stock under the ESPP will be determined by the ESPP Administrator but will not be less than 85%Recommendation of the fair market value of our common stock on the last day of the relevant offering period. The fair market value of a share of our common stock on these measurement dates will be equal to the closing price per share as reported on the NYSE.Board
 |
 |
The Board unanimously recommends a vote “ | ||
| Elanco Animal Health Incorporated |
 |
Proposal No. |
Under our current Articles of Incorporation and Bylaws, directors are seeking shareholder approvalelected by a plurality of the votes cast by shareholders in the election of directors. Under a plurality vote standard, the director nominees who receive the highest number of affirmative votes cast are elected to amendour Board, whether or not votes “For” a director nominee constitute a majority of all votes cast with respect to the nominee and restate our 2018 Elanco Stock Plan, as amended and restated (the “2018 Plan” orregardless of how many shareholders “Withhold” their vote with respect to the “Plan”)nominee. At this Annual Meeting, the existing plurality vote standard will continue to increaseapply.
For Proposal No. 5, the proposed amendments to the Articles of Incorporation would replace the plurality vote standard with a majority vote standard for uncontested elections of directors. Uncontested elections are elections where the number of shares of common stock reserved for issuance under the 2018 Plan by an additional 20,000,000 shares, extend the term of the 2018 Plan, and make design changes to the 2018 Plan. Our continuing ability to offer equity incentive awards under the 2018 Plan is critical to our ability to attract, motivate and retain qualified personnel, particularly as we grow and in light of the highly competitive market for employee talent in which we operate. If shareholders approve this proposal, the Amended and Restated 2018 Elanco Stock Plan (the “Amended 2018 Plan”) will become effective as of the date of shareholder approval. If shareholders dodirector nominees does not approve this proposal, the Amended 2018 Plan described in this proposal will not take effect and our 2018 Plan, as previously amended, will continue to be administered in its current form.
With respect to Proposal No. 5, the proposed amendments to the maximum numberArticles of shares available for issuance under the Plan:
 |
In proposing these amendments to the Articles of Incorporation and seeking to evolve our governance structure, our Board considered shareholder feedback and evolving governance practices. Our Board unanimously concluded, on the recommendation of the Corporate Governance Committee, that the proposed Amended 2018 Plan, which has been included as Appendix C tochanges contemplated by this Proxy Statement.
 |
If this Proposal No. 5 |
| Elanco Animal Health Incorporated | 89 | 2024 Proxy Statement |
This Proposal No. 5 and the amendments to the Articles of Incorporation to adopt a majority vote standard for uncontested elections of directors will be approved if the votes cast “For” the proposal exceed the votes cast “Against” the proposal.
As noted above and below, we are also submitting proposals to amend our Articles of Incorporation to: declassify the Board of Directors (Proposal No. 4), enable shareholders to amend our Bylaws (Proposal No. 6), and enable shareholders to call special meetings of shareholders under certain circumstances (Proposal No. 7). Approval of this Proposal No. 5 is not contingent on approval or disapproval of Proposals No. 4, 6, or 7.
Recommendation of the Board
| The Board unanimously recommends a vote “FOR” the management proposal to amend our Articles of Incorporation to adopt a majority voting provision for uncontested elections of directors. | ||
| Elanco Animal Health Incorporated | 90 | 2024 Proxy Statement |
| Proposal No. 6 Management Proposal to Amend Elanco’s Articles of Incorporation to Provide Shareholders with the Right to Amend our Bylaws |
Under our current Articles of Incorporation, shareholders do not currently have the right to amend our Bylaws.
For Proposal No. 6, the proposed amendments to the Articles of Incorporation would allow for our Bylaws to be amended or repealed, or new Bylaws to be adopted, either by (a) our Board of Directors, by the affirmative vote of at least a majority of the Board of Directors, or (b) our shareholders, at any annual or special meeting of shareholders for which the meeting notice designates that making, amending or repealing provisions of the Bylaws is to be considered, by the affirmative vote of at least a majority of all outstanding shares of the Company’s shareholders. Subjectstock which are entitled to adjustmentvote in the eventelection of specified capitalization events, no more than 26,325,395 shares may be issued pursuantdirectors. Our Board has approved certain conforming changes to our Bylaws, contingent on the effectiveness of these proposed amendments to our Articles of Incorporation.
With respect to Proposal No. 6, the proposed amendments to the exerciseArticles of incentive stock options.
 |
Stock appreciation rights, or “SARs,” typically provide for payments
In proposing these amendments to the holder based upon increases inArticles of Incorporation and seeking to evolve our governance structure, our Board considered shareholder feedback and evolving governance practices. Our Board unanimously concluded, on the pricerecommendation of the Corporate Governance Committee, that the proposed changes contemplated by this Proposal No. 6 should be made.
If this Proposal No. 6 is approved by the requisite vote of our shares fromshareholders at the dateAnnual Meeting, the SAR was grantedproposed amendments to the date thatArticles of Incorporation would become effective upon the right is exercised. The exercise pricefiling of a SAR may not be less thanamended and restated Articles of Incorporation with the fair market valueSecretary of a share on the date of grantState of the SAR. The Committee may elect to settle exercised SARs in cash, in shares, or in a combinationState of cash and shares. UntilIndiana, which we would file promptly following the shares,Annual Meeting if any, are issued, no right to vote or receive dividends or any other rights as a shareholder will exist with respectour shareholders approve the amendments. If this Proposal No. 6 is not approved by the requisite votes of our shareholders at the Annual Meeting, the amendments to the shares subjectArticles of Incorporation described in this Proposal No. 6 would not become effective and shareholders would continue to a SAR, notwithstanding the exercise of the SAR.
This Proposal No. 6 and the underlying sharesamendments to the Articles of Incorporation to adopt a right of shareholders to amend our Bylaws will be approved if the votes cast “For” the proposal exceed the votes cast “Against” the proposal.
As noted above and below, we are also submitting proposals to amend our Articles of Incorporation to: declassify the Board of Directors (Proposal No. 4), change the voting standard for uncontested director elections from a plurality standard to a majority standard (Proposal No. 5), and enable shareholders to call special meetings of shareholders under certain circumstances (Proposal No. 7). Approval of this Proposal No. 6 is not contingent on approval or disapproval of Proposals No. 4, 5, or 7.
| Elanco Animal Health Incorporated | 91 | 2024 Proxy Statement |
Recommendation of the Board
| The Board unanimously recommends a vote “FOR” the management proposal to amend our Articles of Incorporation to provide shareholders the right to amend our Bylaws. | ||
| Elanco Animal Health Incorporated | 92 | 2024 Proxy Statement |
Proposal No. 7 Management Proposal to Amend Elanco’s Articles of Incorporation to Provide Shareholders with the Right to Request Special Meetings of Shareholders |
Under our current Articles of Incorporation and Bylaws, shareholders do not have the right to receive dividends with respectcall special meetings of shareholders.
For Proposal No. 7, the proposed amendments to the underlying shares). The restrictions, ifArticles of Incorporation would allow for special meetings of shareholders to be called (a) at any may be based on, among other conditions, continued service,time by the attainmentBoard of performance conditions,Directors or a combinationthe Chairman of both.
 |
 |
With respect to Proposal No. 7, the proposed amendments to the Articles of Incorporation are included with this proxy statement as Appendix E. This description of the proposed amendments to the Articles of Incorporation is qualified in its entirety by reference to the text of the amendments as set forth in Appendix E.
In proposing these amendments to the Articles of Incorporation and seeking to evolve our governance structure, our Board considered shareholder feedback and evolving governance practices. Our Board unanimously concluded, on the recommendation of the Corporate Governance Committee, that the proposed changes contemplated by this Proposal No. 7 should be made.
| Elanco Animal Health Incorporated | 93 | 2024 Proxy Statement |
If this Proposal No. 7 is approved by the Company’s shareholders. The Plan will continue in effect untilrequisite vote of our shareholders at the tenth anniversaryAnnual Meeting, the proposed amendments to the Articles of Incorporation would become effective upon the filing of amended and restated Articles of Incorporation with the Secretary of State of the date thatState of Indiana, which we would file promptly following the Annual Meeting if our shareholders approve the Amended 2018 Plan, unless earlier terminatedamendments. If this Proposal No. 7 is not approved by our Board. Any awards that are outstanding at the time the Amended 2018 Plan terminates will remain in force according to the terms of the Amended 2018 Plan and the applicable agreement evidencing the award.
This Proposal No. 7 and the amendments to the Articles of Incorporation to adopt a right of shareholders to call special meetings of shareholders will be approved if the votes cast “For” the proposal exceed the votes cast “Against” the proposal.
As noted above, we are also submitting proposals to amend our Articles of Incorporation to: declassify the Board of Directors (Proposal No. 4), change the voting standard for uncontested director elections from a plurality standard to a majority standard (Proposal No. 5), and enable shareholders to amend our Bylaws (Proposal No. 6). Approval of this Proposal No. 7 is not contingent on approval or SAR may be amended to reduce the exercise pricedisapproval of Proposals No. 4, 5, or grant price6.
Recommendation of the shares subject to such option or SAR and (except as permitted under the provisions of the Amended 2018 Plan dealing with certain capitalization adjustments, changes in control, and other events) no option or SAR may be cancelled in exchange for the grant of an option or SAR having a lower per share exercise price or for a cash payment or another award at a time when the option or SAR has a per share exercise price that is higher than the fair market value of the shares.
 |
 |
The Board unanimously recommends a vote “ Incorporation to allow shareholders to request special meetings of shareholders. | ||
| 94 | 2024 Proxy Statement |  |
Stock Ownership Information |
Security Ownership of Directors and Executive Officers
The following table shows the shares of our common stock beneficially owned as of March 20, 2023April 1, 2024 by each director and NEO individually and by all of our executive officers and directors as of such date as a group. Shares reported as beneficially owned include shares held indirectly. It also includes shares subject to stock options exercisable and RSUs subject to conversion in shares of common stock, within sixty60 days of March 20, 2023.April 1, 2024. As of such date, none of these shares were pledged as security.
| Name | Shares Beneficially Owned(1) (#) | Percent of Class (%) | DSUs (#)(2) | | |
| Kapila Anand | 3,200 | * | 69,950 | | |
| John Bilbrey | 41,722 | (3) | * | 74,718 | |
| William Doyle | 15,000 | * | 46,330 | | |
| Art Garcia | 3,525 | * | 59,270 | | |
| Michael Harrington | 16,100 | * | 61,494 | | |
| Paul Herendeen | 10,000 | * | 46,330 | | |
| R. David Hoover | 295,920 | (4) | * | 101,690 | |
| Deborah Kochevar | 1,000 | * | 60,231 | | |
| Lawrence Kurzius | 10,000 | * | 88,456 | | |
| Kirk McDonald | — | * | 60,231 | | |
| Denise Scots-Knight | — | * | 61,218 | | |
| Jeffrey Simmons | 1,469,347 | (5) | * | — | |
| Kathy Turner | — | * | — | ||
| Craig Wallace | — | * | — | ||
| Todd Young | 256,660 | (6) | * | — | |
| Ellen de Brabander | 107,327 | * | — | | |
| Ramiro Cabral | 182,938 | * | — | | |
| Timothy Bettington | 54,044 | (7) | * | — | |
| All directors and executive officers as a group (24 persons) | 2,823,258 | * | 729,918 | |
* Less than 1% of the outstanding shares of common stock.
| (1) | |||||||||
| Includes the following shares not currently outstanding but deemed beneficially owned because of the right to acquire them pursuant to non-qualified stock options currently exercisable or that will become exercisable within 60 |
| (2) | Beneficial ownership excludes shares of DSUs credited to each individual non-employee director’s deferred stock account as of April 1, 2024. Upon termination of the director’s service on the Board, the director will be paid the balance in his or her deferred stock account through the issuance of Elanco common stock, either in a lump sum in January of the second year following the year in which the director leaves the Board or in annual installments over a period between two to 10 years (as selected by the director) beginning at the same time the lump sum payment would be made. The information in this “DSU” column is not required by the rules of the SEC because the DSUs carry no voting rights and director has no right or ability to convert the DSUs to common stock within 60 days of April 1, 2024. Nevertheless, we believe this information provides a more complete picture of the financial stake our directors have in Elanco. |
| (3) | Includes 41,722 shares held indirectly through a |
| (4) | Includes |
| (5) | Includes 45,000 shares held indirectly through a |
| (6) | Includes |
| Includes 54,044 shares not currently outstanding but deemed beneficially owned because of the right to acquire them pursuant to restricted stock units that vest within 60 days. |
| Elanco Animal Health Incorporated | 95 | 2024 Proxy Statement |  |
The following table shows all entities that are the beneficial owners of more than 5% of our common stock as of March 20, 2023.April 1, 2024.
| | Name | | | Number of Shares | | | Percent of Class | |
| | Dodge & Cox(1) | | | 84,775,075 | | | 17.2 % | |
| | The Vanguard Group(2) | | | 45,691,838 | | | 9.3 % | |
| | PRIMECAP Management Company(3) | | | 32,983,562 | | | 6.7 % | |
| | BlackRock, Inc.(4) | | | 30,546,195 | | | 6.2 % | |
| Name | Number of Shares (#) | Percent of Class (%)
| |
| Dodge & Cox(1) | 83,980,900 | 17.00 | |
| PRIMECAP Management Company(2) | 50,600,324 | 10.27 | |
| The Vanguard Group(3) | 49,468,637 | 10.04 | |
| BlackRock, Inc.(4) | 27,781,483 | 5.60 | |
| (1) | As of December 31, |
| (2) | As of December 31, |
| (3) | As of December 31, |
| (4) | As of December 31, 2023, based on information set forth in a Schedule 13G filed with the SEC on January 29, 2024 by BlackRock, Inc. (“BlackRock”). BlackRock’s business address is |
|  |
Delinquent Section 16(a) Reports
Section 16(a) of the Exchange Act requires our directors and executive officers, and persons who own more than 10% of a registered class of our equity securities, to file with the SEC initial reports of ownership and reports of changes in beneficial ownership of such equity securities. To our knowledge, no executive officer or director of Elanco failed to file reports required by Section 16(a) on a timely basis, except as follows due to an administrative oversight: (i) for Mr. Meer, threeone late Form 4 filingsfor Ms. Anand, reporting (a) one RSUthe grant (b) one acquisition of shares from a PSU settlement and one forfeiture of shares for withholding to cover taxes upon vesting, and (c) one RSU grant and two forfeitures of shares to cover taxes upon vesting; and (ii) for Mr. Modi, his initial Form 3 was filed one day late.Deferred Stock Units.
| 96 | 2024 Proxy Statement |  |
Equity Compensation Plan Information |
The following table shows information, as of December 31, 2022,2023, regarding shares of our common stock authorized for issuance under our equity compensation plans. As of such date, other than as described below, no equity securities were authorized for issuance under equity compensation plans not approved by shareholders.
| | | | Number of securities to be issued upon exercise of outstanding options and rights (a) | | | Weighted-average exercise price of outstanding options and rights(1) (b) | | | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (c) | | |
| | Equity Compensation Plans approved by security holders | | | 4,110,324(2) | | | $29.77 | | | 12,691,750 | |
Number of securities to be issued upon exercise of outstanding options and rights (#) (a) | Weighted-average exercise price of outstanding options and rights(1) ($) (b) | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) (#) (c) | | |
| Equity Compensation Plans approved by security holders | 8,019,028(2) | 17.92 | 32,099,362(3) | |
| (1) | The weighted-average exercise price is calculated based solely on the exercise prices of the outstanding stock options and do not reflect the shares that will be issued upon the vesting of outstanding awards of RSUs, which have no exercise price. |
| (2) | This number includes |
| This number includes 26,634,514 shares available for grant under the Amended and Restated 2018 Stock Plan and 5,464,848 shares available for issuance under the Employer Stock Purchase Plan. |
| Elanco Animal Health Incorporated | 97 | 2024 Proxy Statement |  |
General Information About the Annual Meeting |
Online Meeting
We will conduct the Annual Meeting solely via the Internet through a live audio webcast.webcast on May 30, 2024 at 12:00 p.m. Eastern Time. We continue to use the virtual annual meeting format to facilitate shareholder attendance and participation, as we believe this format enables shareholders to participate fully from any location around the world, at no cost to them.
You are entitled to attend and participate in the virtual Annual Meeting only if you held your shares as of the close of business on March 20, 2023April 11, 2024 (the “Record Date”) or if you hold a valid proxy for the Annual Meeting. If you were not an Elanco shareholder as of such date, you may still view the meeting online. Applicable shareholders who wish to participate in the Annual Meeting, or other interested participants who wish to view but not participate in the Annual Meeting, may do so by visitingwww.virtualshareholdermeeting.com/ELAN2024.
www.virtualshareholder
To attend online and participate in the Annual Meeting, shareholders of record will need to use their control number on the Notice of Internet Availability of Proxy Materials (the “Notice of Internet Availability”) provided to them, or their proxy card, to log into www.virtualshareholdermeeting.com/ELAN2023. ELAN2024. If you are a beneficial shareholder and your voting instruction form or Notice of Internet Availability indicates that you may vote those shares through the www.proxyvote.com website, then you may access, participate in and vote at the Annual Meeting with the 16-digit access code indicated on that voting instruction form or Notice of Internet Availability. Otherwise, beneficial shareholders who do not have a control number or access code should contact their bank, broker, or other nominee (preferably at least 5five days before the Annual Meeting) and obtain a “legal proxy” in order to be able to attend, participate in, or vote at the Annual Meeting.
We encourage you to access the meeting prior to the start time. Please allow ample time for online check-in, which will begin at 7:11:45 a.m. Eastern Time. If you have difficulties during the check-in time or during the Annual Meeting, we will have technicians ready to assist you with any difficulties you may have accessing the virtual meeting. If you encounter any difficulties accessing the virtual meeting during the check-in or meeting time, please call the technical support number that will be posted on the Virtual Shareholdervirtual Annual Meeting log-in page.
Shareholders have multiple opportunities to submit questions to us for the Annual Meeting. Shareholders who wish to submit a question in advance may do so at either www.proxyvote.comor on our Annual Meeting website at www.virtualshareholderwww.virtualshareholdermeeting.com/ELAN2024
We reserve the right to eject an attendee or cut off speaking privileges for behavior likely to cause disruption or annoyance or for failure to comply with reasonable requests or the rules of conduct for the meeting, including time limits applicable to attendees who are permitted to speak. We also reserve the right to edit profanity or other inappropriate language and to exclude questions regarding topics that are not pertinent to meeting matters or Elanco business. If we receive substantially similar questions, we may group such questions together and provide a single response to avoid repetition.
| 98 | 2024 Proxy Statement |  |
You are entitled to vote at the Annual Meeting if our records show that you held your shares as of the Record Date. At the close of business on that date, a total of 492,047,948[⋅] shares of our common stock were outstanding and entitled to vote. In addition to shareholders of record of our common stock, “beneficial owners of shares held in street name” as of the Record Date can vote using the methods described below.
| • | Shareholders of Record. If your shares are registered directly in your name with our transfer agent, Computershare Trust Company, N.A., you are the shareholder of record with respect to those shares. |
| • | Beneficial Owners of Shares Held in Street Name. If your shares are held in an account at a bank, broker, or other organization, then you are the “beneficial owner of shares held in street name” (a “beneficial shareholder”). As a beneficial shareholder, you have the right to instruct the person or organization holding your shares how to vote your shares. Most individual shareholders are beneficial owners of shares held in street name. |
Voting Before or During the Annual Meeting
There are four ways to vote:
| • | Online Prior to the Annual Meeting. You may vote by proxy by visiting www.proxyvote.com and entering the control number found on your Notice of Internet Availability. The availability of online voting may depend on the voting procedures of the organization that holds your shares. |
| • | Online During the Annual Meeting. You may vote online during the Annual Meeting by visiting www.virtualshareholder meeting.com/ |
| • | Telephone.If you request printed copies of the proxy materials by mail, you will receive a proxy card or voting instruction form and you may vote by proxy by calling the toll-free number found on the card or form. The availability of telephone voting may depend on the voting procedures of the organization that holds your shares. |
| • | Mail.If you request printed copies of the proxy materials by mail, you will receive a proxy card or voting instruction form and you may vote by proxy by filling out the card or form and returning it in the envelope provided. |
Whether you are a shareholder of record or a beneficial shareholder, you may direct how your shares are voted without participating in the Annual Meeting. We encourage shareholders to vote well before the Annual Meeting, even if they plan to attend the virtual meeting, by completing proxies online or by telephone or, if they received printed copies of these materials, by mailing their proxy cards. The online polls will close at 11:59 p.m. Eastern Time on May 17, 2023.29, 2024.
Shareholders of record may revoke their proxy at any time before the electronic polls close by submitting a later-dated vote online during the Annual Meeting, via the Internet, by telephone, by mail, or by delivering instructions to our Corporate Secretary before the Annual Meeting begins. Beneficial shareholders may revoke any prior voting instructions by contacting the broker, bank, or other nominee that holds their shares or, if applicable, by voting online during the virtual Annual Meeting.
Quorum for the Annual Meeting
In order to have a quorum at the Annual Meeting, holders of a majority of the outstanding shares entitled to vote at the Annual Meeting must be present or represented by proxy for the transaction of business. Your shares will be counted for purposes of determining if there is a quorum if you are entitled to vote and you are present at the Annual Meeting, or if you have properly voted by proxy online, by phone, or by submitting a proxy card or voting instruction form by mail prior to the Annual Meeting.
Broker non-votes (as described below) and abstentions are counted for purposes of determining whether a quorum is present. If a quorum is not present, we may propose to adjourn the Annual Meeting and reconvene the Annual Meeting at a later date.
| 99 | 2024 Proxy Statement |  |
Voting Standards
Each share of common stock is entitled to one vote at the Annual Meeting. ToFor Proposal No. 1, to be elected, (Proposal No. 1), the director nominees must receive a plurality of the votes cast by the holders of shares entitled to vote in the election at the meeting,Annual Meeting, provided a quorum is present. A “plurality of the votes cast” means that the individuals with the highest number of “for”“For” votes are elected as directors up to the maximum number of directors to be elected. “Votes cast” excludes abstentions and any broker non-votes. Accordingly, abstentions and broker non-votes will have no effect on the election of directors. Brokers do not have discretionary authority with respect to the election of directors.
For Proposal Nos. 2, 3, 45, 6 and 5,7, each proposal will be approved if the votes cast for the proposal exceed those cast against the proposal at the meeting. Abstentions or broker non-votes with respect to these proposals, if any, will have no effect on the outcome of the vote. Brokers have discretionary authority with respect to Proposal No. 2. Brokers do not have discretionary authority with respect to Proposals Nos. 1, 3, 5, 6 and 7.
For Proposal No. 4, the affirmative vote of at least a majority of all of the outstanding shares of our common stock, voting together as a single class, is required to approve the proposal. Abstentions and 5.broker non-votes with respect to Proposal No. 4 will have the same effect as a vote against the proposal. Brokers do not have discretionary authority with respect to Proposal No. 4.
The following chart describes the proposals to be considered at the Annual Meeting, the vote required to elect directors and to adopt each other proposal and the manner in which votes will be counted.
Voting Options | Vote Required to Adopt the Proposal | Effect of Abstentions | Effect of “Broker Non-Votes” | |||||||||||
No. 1 – Election of Directors | For, against or abstain on each nominee. | Plurality of votes cast. | No effect. | No effect. No broker discretion to vote. | ||||||||||
No. 2 – Ratification of Independent Auditor | For, against or abstain. | More votes | No effect. | Brokers have discretion to vote. | ||||||||||
No. 3 – Advisory Vote on Executive Compensation | For, against or abstain. | More votes | No effect. | No effect. No broker discretion to vote. | ||||||||||
No. 4 – | For, against or abstain. | Against. No broker discretion to vote. | ||||||||||||
| No. 5 – Management Proposal to Amend Elanco's Articles of Incorporation to Adopt a Majority Vote Standard for Uncontested Elections of Directors | For, against or abstain. | More votes | No effect. | No effect. No broker discretion to vote. | ||||||||||
For, against or abstain. | More votes | No effect. | No effect. No broker discretion to vote. | |||||||||||
| No. 7 – Management Proposal to Amend Elanco's Articles of Incorporation to Provide Shareholders With the Right to Request Special Meetings of Shareholders | More votes “For” than “Against.” | No effect. | No effect. No broker discretion to vote. |
| Elanco Animal Health Incorporated | 100 | 2024 Proxy Statement |
If you complete and submit your proxy voting instructions, the individuals named as proxies will follow your instructions. If you are a shareholder of record and you submit proxy voting instructions but do not direct how to vote on each item, the individuals named as proxies will vote as our Board recommends on each proposal and as they may determine in their best judgment with respect to any other matters properly presented for a vote at the Annual Meeting.
If you are a beneficial shareholder and do not provide the broker that holds your shares with specific voting instructions, then such broker may generally vote your shares in their discretion on “routine” matters but cannot vote on “non-routine matters.” For the Annual Meeting, only Proposal No. 2 is considered a routine matter.
 |
Cost of Proxy Solicitation
We are providing these proxy materials in connection with the solicitation by our Board of proxies to be voted on at the Annual Meeting. We will pay the cost of this proxy solicitation. In addition to soliciting proxies by mail, we expect that a number of our employees will solicit shareholders personally, electronically and by telephone. None of these employees will receive any additional compensation for doing this. We have retained Innisfree M&A Incorporated to assist in the solicitation of proxies for a fee of $20,000$50,000 plus reimbursement of expenses. We will, on request, reimburse brokers, banks and other nominees for their expenses in sending proxy materials to their customers who are beneficial shareholders and obtaining their voting instructions.
Shareholder List
Our list of shareholders as of the Record Date will be available for inspection for five business days prior to the Annual Meeting. If you want to inspect the shareholder list, please contact our Investor Relations department at investor@elanco.com to schedule an appointment. In addition, the list of shareholders will also be available during the Annual Meeting through the meeting website for those shareholders who choose to attend.
| 101 | 2024 Proxy Statement |  |
Submission of Proposals or Nominations |
Rule 14a-8 Proposals for Inclusion in the Proxy Statement for the 20242025 Annual Meeting
Pursuant to Rule 14a-8 under the Exchange Act (“Rule 14a-8”), some shareholder proposals may be eligible for inclusion in our Proxy Statement for the 20242025 Annual Meeting. These proposals must be received by our Corporate Secretary at our principal offices, through one of the means discussed in the “Communicating withWith Us” section below, by no later than the close of business (5:00 p.m. Eastern Time) on December 8, 2023.20, 2024. Proposals submitted for inclusion in our Proxy Statement for the 20242025 Annual Meeting must comply with all requirements of Rule 14a-8.
Director Nominations (Including Proxy Access Nominations) or Other Proposals for Presentation at the 20242025 Annual Meeting
. PROXY ACCESS
We have adopted proxy access, which permits a shareholder, or group of up to 20 shareholders, owning 3% or more of our outstanding common stock continuously for at least three years, to submit director nominations for up to two individuals or 20% of our Board (whichever is greater) for inclusion in our Proxy Statement if the shareholder(s) and the nominee(s) meet the requirements in our Bylaws.
DIRECTOR NOMINATIONS AND OTHER PROPOSALS, OTHER THAN PROXY ACCESS AND RULE 14a-8
Under our Bylaws, a shareholder may nominate a candidate for election to our Board (other than pursuant to the proxy access provisions of our Bylaws) or may propose any business for presentation at the 20242025 Annual Meeting (other than proposals presented under Rule 14a-8) pursuant to the advance notice provisions of the Bylaws.
DEADLINE FOR NOTICE
A shareholder who desires to nominate a candidate for election to our Board (whether pursuant to the proxy access provisions of our Bylaws or otherwise) or to propose any business for presentation at the 20242025 Annual Meeting (other than proposals presented under Rule 14a-8) pursuant to the advance notice provisions of the Bylaws, must give notice to our Corporate Secretary at our principal executive offices, through one of the means discussed in the “Communicating withWith Us” section below, by no earlier than January 18, 202430, 2025 and no later than the close of business (5:00 p.m. Eastern Time) on February 17, 2024.March 1, 2025. The notice must include the information specified in our Bylaws, including information concerning the nominee or proposal (as applicable) and information about the shareholder’s ownership of and agreements related to our stock.
| Elanco Animal Health Incorporated | 102 | 2024 Proxy Statement |
If the 20242025 Annual Meeting is advanced or delayed more than 30 days from the anniversary of the Annual Meeting, a shareholder seeking to nominate a candidate for election to our Board or propose any business at our 20242025 Annual Meeting pursuant to the advance notice provisions of the Bylaws must submit notice of any such nomination and of any such proposal that is not made pursuant to Rule 14a-8 by the close of business (5:00 p.m. Eastern Time) on the later of 120 days in advance of the 20242025 Annual Meeting or, if later, 10 days following the date on which public disclosure of the date of the meeting was first made.
In addition to satisfying the foregoing requirements, to comply with the universal proxy rules, shareholders who intend to solicit proxies in support of director nominees other than the Board’s nominees must provide notice that sets forth the information required by Rule 14a-19 under the Securities Exchange Act of 1934 no later than March 24, 2024.31, 2025.
REQUIREMENTS FOR DIRECTOR NOMINEES AND OTHER PROPOSALS
All director nominations and shareholder proposals must comply with the requirements of our Bylaws, including, with respect to director nominations, the eligibility requirements contained therein. A copy of our Bylaws is available on our website at www.elanco.com/en-us/about-us/governance/corporate by clicking on the “Bylaws” link. The Chairman may refuse to acknowledge or introduce any such matter at the 20242025 Annual Meeting if notice of the matter is not received within the applicable deadlines or does not comply with our Bylaws. If a shareholder does not meet these deadlines or does not satisfy the requirements of Rule 14a-4 of the Exchange Act, the persons named as proxies will be allowed to use their discretionary voting authority when and if the matter is raised at the 20242025 Annual Meeting.
| 103 | 2024 Proxy Statement |  |
Other Information |
Communicating withWith Us
To communicate with our Board (or any individual member), make a proposal or director nomination, introduce business at an annual meeting of shareholders, revoke a prior proxy instruction, or request copies of our governance-related documents, please contact us via e-mail to elanco_corporate_secretary@elancoah.com or by mail to Elanco Animal Health Incorporated, 2500 Innovation Way, Greenfield, IN 46140, Attention: Corporate Secretary.
The Corporate Secretary regularly forwards to the addressee all correspondence other than mass mailings, advertisements and other materials not relevant to our business. However, we reserve the right not to forward to Board members any abusive, threatening, or otherwise inappropriate materials.
Notice of Internet Availability
We use the Internet as the primary means of furnishing proxy materials to shareholders. We are sending a Notice of Internet Availability to our shareholders with instructions on how to access the proxy materials online at www.proxyvote.comor request a printed copy of the materials. Our proxy materials are also available at https://investor.elanco.com/financials/annual-reports/annual-reports/.
Shareholders may follow the instructions in the Notice of Internet Availability to elect to receive future proxy materials in print by mail or electronically by e-mail. We encourage shareholders to take advantage of the availability of the proxy materials online to help reduce the environmental impact of our annual meetings and reduce our printing and mailing costs.
Householding of Proxy Materials
The SEC has adopted rules that permit companies and intermediaries (such as brokers and banks) to satisfy the delivery requirements for Proxy Statements with respect to two or more security holders sharing the same address by delivering a single Notice of Internet Availability or Proxy Statement addressed to those security holders. This process, which is commonly referred to as “householding,” potentially provides extra convenience for security holders and cost savings for companies.
Several brokers and banks with accountholders who are Elanco shareholders will be “householding” our proxy materials. As indicated in the notice provided by these brokers to Elanco shareholders, a single Proxy Statementproxy statement will be delivered to multiple shareholders sharing an address unless contrary instructions have been received from an affected shareholder. Once you have received notice from your broker that it will be “householding” communications to your address, “householding” will continue until you are notified otherwise or until you revoke your consent. If, at any time, you no longer wish to participate in “householding” and you prefer to receive a separate proxy statement, please notify your broker, contact Broadridge Financial Solutions at 1-866-540-7095 or by writing to Broadridge, Householding Department, 51 Mercedes Way, Edgewood, New York 11717, or send a written request to Elanco Animal Health Incorporated, 2500 Innovation Way, Greenfield, IN 46140, Attention: Investor Relations or via e-mail at investor@elanco.com. Following this request, we will undertake to deliver promptly a separate copy of the proxy materials. Shareholders who currently receive multiple copies of the Proxy Statement at their address and would like to request “householding” of their communications should contact their broker or bank.
| 104 | 2024 Proxy Statement |  |
This document includesProxy Statement contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995,federal securities laws, including, without limitation, statements concerning future business activities and financial performance. Forward-looking statements are based on our current expectations and assumptions regarding our long-term financial targetsbusiness, the economy and ESG goalsother future conditions and strategies. These statements involveare subject to inherent uncertainties, risks and uncertainties. Actualchanges in circumstances that are difficult to predict. As a result, our actual results couldmay differ materially from any future results expressed or impliedthose contemplated by the forward-looking statements. Important risk factors that could cause actual results to differ materially from those in the forward-looking statements for a variety of reasons, including dueinclude but are not limited to the following: operating in a highly competitive industry; the success of our R&D and licensing efforts; the impact of disruptive innovations; competition from generic products; an outbreak of infectious disease carried by farm animals; risks related to the evaluation of animals; consolidation of our customers and uncertainties that are discusseddistributors; the impact of increased or decreased sales into our distribution channels; our dependence on the success of our top products; our ability to complete acquisitions and divestitures and successfully integrate the businesses we acquire, including the proposed divestiture of our aqua business; our ability to implement our business strategies or achieve targeted cost efficiencies and gross margin improvements; manufacturing problems and capacity imbalances; fluctuations in inventory levels in our most recently filed periodic reportsdistribution channels; the impact of weather conditions, including those related to climate change, and the availability of natural resources; the effects of a human disease outbreak, epidemic, pandemic or other widespread public health concern; the loss of key personnel or highly skilled employees; adverse effects of labor disputes, strikes and/or work stoppages; the effect of our substantial indebtedness on our business; changes in interest rates; risks related to the write-down of goodwill or identifiable intangible assets; the lack of availability or significant increases in the cost of raw materials; risks related to our presence in foreign markets; risks related to currency rate fluctuations; risks related to underfunded pension plan liabilities; the potential impact that actions by activist shareholders could have on the pursuit of our business strategies; risks related to certain governance provisions in our constituent documents; actions by regulatory bodies, including as a result of their interpretation of studies on product safety; the possible slowing or cessation of acceptance and/or adoption of our farm animal sustainability initiatives; the impact of increased regulation or decreased governmental financial support related to the raising, processing or consumption of farm animals; the impact of litigation, regulatory investigations, and other legal matters, including the risk to our reputation; challenges to our intellectual property rights or our alleged violation of rights of others; misuse, off-label or counterfeiting use of our products; unanticipated safety, quality or efficacy concerns and the impact of identified concerns associated with our products. For additional information about these and other factors that could cause actual results to differ materially from forward-looking statements, please see the Risk Factors in our latest Form 10-K and Form 10-Q and subsequent filings.10-Qs filed with the SEC. Any forward-looking statement made by us in this Proxy Statement speaks only as of the date hereof. We assumeundertake no obligation to publicly update or to revise any forward-looking statementsstatement, whether as a result of new information, future developments or information, which speak onlyotherwise, except as of their respective dates.may be required by law.
Website references and their hyperlinks throughout this document are provided for convenience only and the content on the referenced websites, including but not limited to the content contained in our 20212023 ESG Summary Report, is not incorporated herein by reference into this Proxy Statement, nor does it constitute a part of this Proxy Statement.
Financial Matters
Our financial statements for the year ended December 31, 20222023 are included in our 20222023 Annual Report, which we provide to our shareholders at the same time as this Proxy Statement. Our 20222023 Annual Report and this Proxy Statement are also posted on our website at https://investor.elanco.com/financials/annual-reports/. If you have not received or do not have access to the 20222023 Annual Report, please send a written request to Elanco Animal Health Incorporated, 2500 Innovation Way, Greenfield, IN 46140, Attention: Investor Relations.
| Elanco Animal Health Incorporated | 105 | 2024 Proxy Statement |
Matters to be Presented
We know of no other matters to be submitted to shareholders at the Annual Meeting, other than the proposals identified in this Proxy Statement. If any other matters properly come before shareholders at the Annual Meeting, it is the intention of the persons named on the proxy to vote the shares represented thereby on such matters in accordance with their best judgment. If the meeting is adjourned or postponed, the persons named on the proxy can vote such shares at the adjournment or postponement as well.
By orderOrder of the Board of Directors,
Shiv O'Neill
Executive Vice President, General Counsel
and
| 106 | 2024 Proxy Statement |  |
Appendix A Reconciliation of GAAP Reported to Selected Non-GAAP Adjusted Information |
In this Proxy Statement, we use non-GAAP financial measures such as EBITDA, adjusted EBITDA, adjusted EBITDA margin adjusted gross margin and net debt.
We believe these non-GAAP financial measures are useful to investors because they provide greater transparency regarding our operating performance. Reconciliation of non-GAAP financial measures and reported GAAPU.S. generally accepted accounting principles (GAAP) financial measures are included in the tables accompanying this Appendix Apress release and are posted on our website at www.elanco.com.www.elanco.com. The primary material limitations associated with the use of such non-GAAP measures as compared to U.S. GAAP results include the following: (i) they may not be comparable to similarly titled measures used by other companies, including those in our industry, (ii) they exclude financial information and events, such as the effects of an acquisition or amortization of intangible assets, that some may consider important in evaluating our performance, value or prospects for the future, (iii) they exclude items or types of items that may continue to occur from period to period in the future and (iv) they may not exclude all unusual or non-recurring items, which could increase or decrease these measures, which investors may consider to be unrelated to our long-term operations. These non-GAAP measures are not, and should not, be viewed as substitutes for U.S. GAAP reported measures. We encourage investors to review our unaudited condensed consolidated and combined financial statements in their entirety and caution investors to use U.S. GAAP measures as the primary means of evaluating our performance, value and prospects for the future, and non-GAAP measures as supplemental measures.
 |
Adjusted Net IncomeEPS
We define adjusted net income as net income (loss) excluding amortization of intangible assets, purchase accounting adjustments to inventory, integration costs of acquisitions, severance, goodwill and other asset impairment,impairments, gain on sale of assets, facility exit costs, tax valuation allowances and other specified significant items, such as unusual or non-recurring items that are unrelated to our long-term operations adjusted for income tax expense associated with the excluded financial items.
| Elanco Animal Health Incorporated | A-1 | 2024 Proxy Statement |
We define adjusted EPS as adjusted net income divided by the number of weighted-average shares outstanding for the periods ended December 31, 2023 and 2022. The following is a reconciliation of GAAP Revisedreported to selected non-GAAP adjusted information, which includes GAAP net income (loss) to adjusted net income and GAAP EPS to adjusted EPS for the yeartwelve months ended December 31, 20222023 and 2021 to Selected Non-GAAP Adjusted information:
| | | | 2022 | | | 2021 | | |||||||||||||
| | GAAP Revised(a) | | | Adjusted Items(c) | | | Non- GAAP(b) | | | GAAP Revised(a) | | | Adjusted Item(c) | | | Non- GAAP(b) | | |||
| | Cost of sales(1) | | | $1,913 | | | $— | | | $1,913 | | | $2,132 | | | $64 | | | $2,068 | |
| | Amortization of intangible assets | | | $528 | | | $528 | | | $— | | | $556 | | | $556 | | | $— | |
| | Asset impairment, restructuring and other special charges(2)(3) | | | $183 | | | $183 | | | $— | | | $634 | | | $634 | | | $— | |
| | Interest expense, net of capitalized interest(4) | | | $241 | | | $20 | | | $221 | | | $236 | | | $— | | | $236 | |
| | Other (income) expense, net(5)(6) | | | $32 | | | $2 | | | $30 | | | $5 | | | $(14) | | | $19 | |
| | Income (loss) before taxes | | | $(72) | | | $733 | | | $662 | | | $(571) | | | $1,240 | | | $669 | |
| | Provision for taxes(7)(8) | | | $6 | | | $(111) | | | $117 | | | $(88) | | | $(236) | | | $147 | |
| | Net income (loss) | | | $(78) | | | $622 | | | $544 | | | $(483) | | | $1,004 | | | $522 | |
| | Earnings (loss) per share: | | | | | | | | | | | | | | ||||||
| | basic | | | $(0.16) | | | $1.27 | | | $1.11 | | | $(0.99) | | | $2.06 | | | $1.07 | |
| | diluted | | | $(0.16) | | | $1.26 | | | $1.11 | | | $(0.99) | | | $2.06 | | | $1.07 | |
| | Adjusted weighted average shares outstanding : | | | | | | | | | | | | | | ||||||
| | basic | | | 488.3 | | | 488.3 | | | 488.3 | | | 487.2 | | | 487.2 | | | 487.2 | |
| | diluted(9) | | | 488.3 | | | 492.2 | | | 492.2 | | | 487.2 | | | 488.9 | | | 488.9 | |
Twelve months ended December 31, 2023 | Twelve months ended December 31, 2022 | |||||||||||||||||||||||
| $ in millions, expect per share amounts | GAAP Reported | Adjusted Items (b) | Non- GAAP (a) | GAAP Reported | Adjusted Items (b) | Non- GAAP (a) | ||||||||||||||||||
| Cost of sales (1) | $ | 1,931 | $ | 2 | $ | 1,929 | $ | 1,913 | $ | — | $ | 1,913 | ||||||||||||
| Amortization of intangible assets | $ | 548 | $ | 548 | $ | — | $ | 528 | $ | 528 | $ | — | ||||||||||||
| Asset impairment, restructuring and other special charges (2) | $ | 127 | $ | 127 | $ | — | $ | 183 | $ | 183 | $ | — | ||||||||||||
| Goodwill impairment | $ | 1,042 | $ | 1,042 | $ | — | $ | — | $ | — | $ | — | ||||||||||||
| Interest expense, net of capitalized interest (3) | $ | 277 | $ | — | $ | 277 | $ | 241 | $ | 20 | $ | 221 | ||||||||||||
| Other expense, net (4) | $ | 75 | $ | 42 | $ | 33 | $ | 32 | $ | 2 | $ | 30 | ||||||||||||
| (Loss) income before taxes | $ | (1,195 | ) | $ | 1,761 | $ | 566 | $ | (72 | ) | $ | 733 | $ | 661 | ||||||||||
| Income tax expense (5) | $ | 36 | $ | (91 | ) | $ | 127 | $ | 6 | $ | (111 | ) | $ | 117 | ||||||||||
| Net (loss) income | $ | (1,231 | ) | $ | 1,670 | $ | 439 | $ | (78 | ) | $ | 622 | $ | 544 | ||||||||||
| (Loss) earnings per share: | ||||||||||||||||||||||||
| basic | $ | (2.50 | ) | $ | 3.39 | $ | 0.89 | $ | (0.16) | $ | 1.27 | $ | 1.11 | |||||||||||
| diluted | $ | (2.50 | ) | $ | 3.39 | $ | 0.89 | $ | (0.16) | $ | 1.26 | $ | 1.11 | |||||||||||
| Adjusted weighted average shares outstanding: | ||||||||||||||||||||||||
| basic | 492.3 | 492.3 | 492.3 | 488.3 | 488.3 | 488.3 | ||||||||||||||||||
| diluted (6) | 492.3 | 493.7 | 493.7 | 488.3 | 492.2 | 492.2 | ||||||||||||||||||
Numbers may not add due to rounding.
The table above reflects only line items with non-GAAP adjustments.
| (a) |
| The company uses non-GAAP financial measures that differ from financial statements reported in conformity with |
| (b) | Adjustments to |
| (1) |
| (2) |
Elanco Animal Health Incorporated | A-2 |  |
| Adjustments of $20 million for the year ended December 31, 2022, related to |
| (4) | Adjustments of $42 million for the year ended December 31, 2023, primarily related to settlement provisions recorded in 2023 related to the Seresto class action lawsuits ($15 million) and for a possible resolution or settlement relating to a previously disclosed matter with the SEC ($12.5 million), as well as a write-off of an acquisition-related tax indemnification receivable from Bayer ($10 million) and the impact of hyperinflationary accounting in Turkey ($7 million), partially offset by decreases in contingent consideration payable to NutriQuest ($4 million). Adjustments of $2 million for the year ended December 31, 2022, |
| (5) |
| (6) |
| During the years ended December 31, |
| | | | 2020 | | |||||||
| | | | GAAP Revised(a) | | | Adjusted Items(c) | | | Non- GAAP(b) | | |
| | Cost of sales(1) | | | $1,667 | | | $96 | | | $1,571 | |
| | Amortization of intangible assets | | | $360 | | | $360 | | | $— | |
| | Asset impairment, restructuring and other special charges(2) | | | $623 | | | $623 | | | $— | |
| | Interest expense, net of capitalized interest(3) | | | $150 | | | $3 | | | $147 | |
| | Other income, net(4) | | | $(178) | | | $(169) | | | $(9) | |
| | Income (loss) before taxes | | | $(677) | | | $914 | | | $237 | |
| | Provision for taxes(5) | | | $(103) | | | $(142) | | | $39 | |
| | Net income (loss) | | | $(574) | | | $773 | | | $199 | |
| | Earnings (loss) per share: | | | | | | | | |||
| | basic | | | $(1.30) | | | $1.75 | | | $0.45 | |
| | diluted | | | $(1.30) | | | $1.75 | | | $0.45 | |
| | Adjusted weighted average shares outstanding: | | | | | | | | |||
| | basic | | | 441.4 | | | 441.4 | | | 441.4 | |
| | diluted(6) | | | 441.4 | | | 442.6 | | | 442.6 | |
 |
| Elanco Animal Health Incorporated | A-3 | 2024 Proxy Statement |
Adjusted EBITDA / Adjusted EBITDA Margin
We define adjusted EBITDA as net income (loss) adjusted for interest expense (income), which includes debt extinguishment losses, income tax expense (benefit), tax valuation allowances and depreciation and amortization, further adjusted to exclude purchase accounting adjustments to inventory, integration costs of acquisitions, severance, goodwill and other asset impairment, gainimpairments, gains on sale of assets, facility exit costs and other specified significant items, such as unusual or non-recurring items that are unrelated to our long-term operations adjustedoperations.
For the periods presented, we have not made adjustments for income tax expense associated with the excluded financial items.all items that may be considered unrelated to our long-term operations. We definebelieve adjusted EBITDA, margin aswhen used in conjunction with our results presented in accordance with GAAP and its reconciliation to net income (loss), enhances investors’ understanding of our performance, valuation and prospects for the future. We also believe adjusted EBITDA dividedis a measure used in the animal health industry by total revenue.analysts as a valuable performance metric for investors. The following is a reconciliation of U.S. GAAP Net Lossnet income (loss) for the twelve months ended December 31, 2023 and 2022, to EBITDA, adjusted EBITDA and adjusted EBITDA and a calculation ofMargin, which is adjusted EBITDA margindivided by total revenue, for the year ended December 31, 2022 and 2021.
 |
| | | | Year Ended December 31, | | ||||
| | | | 2022 | | | 2021 | | |
| | Revised net loss(1) | | | $(78) | | | $(483) | |
| | Net interest expense | | | 241 | | | 236 | |
| | Income tax expense (benefit) | | | 6 | | | (88) | |
| | Depreciation and amortization | | | 682 | | | 716 | |
| | EBITDA | | | $851 | | | $381 | |
| | Non-GAAP Adjustments: | | | | | | ||
| | Cost of sales | | | $— | | | $64 | |
| | Asset impairment, restructuring and other special charges | | | 183 | | | 634 | |
| | Accelerated depreciation(2) | | | (19) | | | (6) | |
| | Other (income) expense, net | | | 2 | | | (14) | |
| | Adjusted EBITDA | | | $ 1,017 | | | $ 1,059 | |
| | Adjusted EBITDA Margin | | | 23.1% | | | 22.2% | |
| Twelve Months Ended December 31, | ||||||||
| $ in millions | 2023 | 2022 | ||||||
| Reported net loss | $ | (1,231 | ) | $ | (78 | ) | ||
| Net interest expense | 277 | 241 | ||||||
| Income tax expense (benefit) | 36 | 6 | ||||||
| Depreciation and amortization | 694 | 682 | ||||||
| EBITDA | $ | (224 | ) | $ | 851 | |||
| Non-GAAP Adjustments: | ||||||||
| Cost of sales | $ | 2 | $ | — | ||||
| Asset impairment, restructuring and other special charges | 127 | 183 | ||||||
| Goodwill impairment | 1,042 | — | ||||||
| Other expense, net | 42 | 2 | ||||||
| Accelerated depreciation and amortization(1) | (10 | ) | (19 | ) | ||||
| Adjusted EBITDA | $ | 979 | $ | 1,017 | ||||
| Adjusted EBITDA Margin | 22.2 | % | 23.1 | % | ||||
Numbers may not add due to rounding.
| (1) |
| Represents depreciation and amortization of certain assets that was accelerated during the periods presented. These assets became fully depreciated and amortized during the second quarter of 2023. This amount must be added back to arrive at |
| Elanco Animal Health Incorporated | A-4 | 2024 Proxy Statement |
Net Debt
We define net debt as gross debt less cash and cash equivalents on ourthe balance sheet. We define gross debt as the sum of the current portion of long-term debt and long-term debt excluding unamortized debt issuance costs. We define the net debt leverage ratio as gross debt less cash and cash equivalents divided by adjusted EBITDA. This calculation does not include Term Loan B covenant-related adjustments that reduce this leverage ratio.
The following is a reconciliation of gross debt to net debt for the year endedas of December 31, 2022:2023:
| $ in millions | ||||
| Long-term debt | $ | 5,736 | ||
| Current portion of long-term debt | 38 | |||
| Less: Unamortized debt issuance costs | (50 | ) | ||
| Total gross debt | 5,824 | |||
| Less: Cash and cash equivalents | 352 | |||
| Net Debt | $ | 5,472 | ||
| Net Debt Leverage Ratio | 5.6 | x | ||
 |
Elanco Animal Health Incorporated | 2024 Proxy Statement |
Appendix B Proposed Amendments to Articles of Incorporation (Proposal No. 4) | ||
The proposed amendments to Article 9 of our Articles of Incorporation related to Proposal No. 4 are shown below. Additions are indicated by underlining and deletions are indicated by strike-outs. The full text of our Articles of Incorporation can be found on our website at www.elanco.com/en-us/about-us/governance/corporateby clicking on the “Articles of Incorporation” link.
| 9. | The following provisions are inserted for the |
| a. | The number of directors of the Corporation, exclusive of directors who may be elected by the holders of any one or more series of Preferred Stock pursuant to Article 9(b) (the “Preferred Stock Directors”), shall not be less than five, the |
| b. |
 |
 |
 |
 |
 |
 |
| c. |
|  |
| d. |
 |
|  |
| e. |
 |
| 2024 Proxy Statement |
Appendix C Proposed Amendments to (Proposal No. 5) | ||
The proposed amendments to Article 13 of our Articles of Incorporation related to Proposal No. 5 are shown below. Additions are indicated by underlining and deletions are indicated by strike-outs. The full text of our Articles of Incorporation can be found on our website at www.elanco.com/en-us/about-us/governance/corporate by clicking on the “Articles of Incorporation” link.
| 13. |
|  |
| C-1 | 2024 Proxy Statement |
Appendix D Proposed Amendments to Articles of Incorporation (Proposal No. 6) | ||
The proposed addition of Article 14 to our Articles of Incorporation related to Proposal No. 6 is shown below. The full text of our Articles of Incorporation can be found on our website at www.elanco.com/en-us/about-us/governance/corporate by clicking on the “Articles of Incorporation” link.
| 14. | Except as otherwise expressly provided by the IBCL or these Amended and |
 |
| Elanco Animal Health Incorporated | D-1 | 2024 Proxy Statement |
Appendix E Proposed Amendments to Articles of Incorporation (Proposal No. 7) | ||
The proposed addition of Article 15 to our Articles of Incorporation related to Proposal No. 7 is shown below. The full text of our Articles of Incorporation can be found on our website at www.elanco.com/en-us/about-us/governance/corporate by clicking on the “Articles of Incorporation” link.
| 15. | Special meetings of shareholders may be called (a) at any time by the Board of Directors or the Chairman of the |
 |
 |
 |  |
 |

 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |